Laboratory Evaluation of the Efficacy of Chlorine Dioxide (ClO2) Composite Powder for Remediation of Petroleum Hydrocarbon-Contaminated Soils
Roger Saint-Fort* and Avneet Kaur
Department of Environmental Science, Mount Royal University, Canada
Submission: May 01, 2024; Published: May 20, 2024
*Corresponding author: Roger Saint-Fort, Department of Environmental Science, Faculty of Science & Technology, Mount Royal University, 4825 Mount Royal Gate SW, Calgary, AB, T3E6K6, Canada; Email: rsaintfort@mtroyal.ca
How to cite this article: Roger Saint-F, Avneet K. Laboratory Evaluation of the Efficacy of Chlorine Dioxide (ClO2) Composite Powder for Remediation of Petroleum Hydrocarbon-Contaminated Soils. Int J Environ Sci Nat Res. 2024; 33(3): 556367. DOI: 10.19080/IJESNR.2024.33.556367
Abstract
The evaluation of efficacy of ClO2 composite powder as a chemical oxidant for remediating petroleum hydrocarbon in soils has been explored. The main purpose of this study is to investigate the oxidative degradation capacity of and the resulting effects on soil chemistry. The most economical dosage was a ratio of 1g product:27g of contaminated soil. Destruction efficiency in the samples ranged from 7 to 41%. Decrease in soil samples pH ranged from 0.08 to 0.80 and 0.04 to 1.01, respectively. Increasing trend in hydrocarbon degradation was F1≈F2≈F3>F4. The degradation mechanism of the hydrocarbon constituents was postulated to occur in two stages. The first fast-reaction stage involves ClO2 (aq) in initial electron transfer and in the second-stage, the generation of hypochlorous acid (HOCl) and itself partially dissociates, forming hypochlorite anion, ClO−, which would recurrently react with the soil contaminants. SAR values remained unchanged relative unchanged. 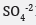 level increases in all samples due to oxidative transformations of sulfur containing compounds by ClO2. Soil EC rises as ClO2 dosage increased and was attributed to higher levels of Cl- and
level increases in all samples due to oxidative transformations of sulfur containing compounds by ClO2. Soil EC rises as ClO2 dosage increased and was attributed to higher levels of Cl- and 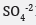 . As a direct benefit,
. As a direct benefit, 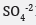 provides a source of nutrient fertilizer in the soil for plants.
provides a source of nutrient fertilizer in the soil for plants.
Keywords: Contamination; Hydrocarbon; Chlorine dioxide
Introduction
Anthropogenic activities and mismanagement can often lead to spilling of petroleum hydrocarbons in the soil, with real risks to human health and negative affect on the soil ecosystem [1-3]. In addition, the spillage can give rise to unwanted and uncontrollable migration of petroleum hydrocarbon substances in the air, surface and ground water bodies. Petroleum represents one of the most important sources of energy and a raw material in the chemical and manufacture industries. From a strict chemical perspective, the term petroleum hydrocarbons refer to a complex mixture of organic compounds consisting predominantly of hydrogen and carbon atoms, and containing small quantities of oxygen, nitrogen, sulfur as well as trace of metallic constituents. The impacts of petroleum hydrocarbons on environmental ecosystems have been examined and documented [4-6], including unwanted changes to soil physicochemical properties, toxicity to biota, recalcitrancy to natural degradation, bioaccumulation, degradation of water and air quality, destruction of flora, exposure to airborne contaminants, loss of productive land and economic activities [7-9]. As such, the predicament of petroleum hydrocarbons contaminated soils and their remediation is among the most urgent and complex tasks facing many countries around the world in terms of environmental protection and financial aspects.
Petroleum hydrocarbons is regarded as the most recurring environmental contaminant. Scientists have recognized the potential dangers of petroleum hydrocarbons accidental released on land and devoted to the development of various remediation technologies. The soil remediation approaches available can be classified as in-situ or ex-situ while falling under the treatment category of chemical, physical, biological, electrochemical, or thermal treatments. However, the relative success of these various remediation methods will depend mainly on their large-scale applicability in the field and low-cost effectiveness.
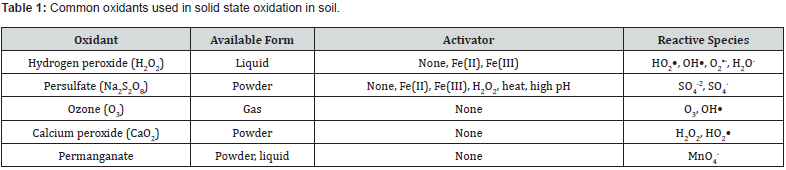
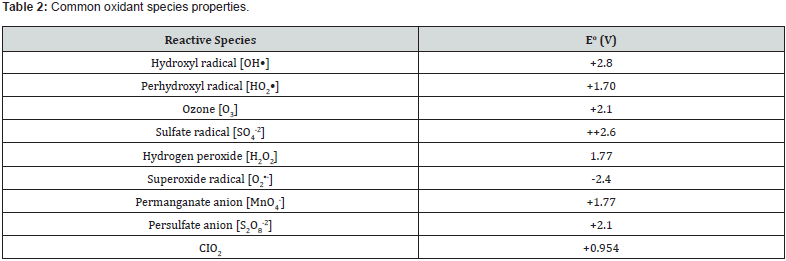
Interactions between soil-hydrocarbon contaminant tend to be highly complex. Consequently, affected by hydrocarbon constituents and characteristics, soil solution surface tension, soil mineralogy, biological, chemical and physical properties, soil aqueous regime, extent of sorption and desorption processes, nature of mechanism of the soil-contaminant interactions as well as contaminant mobilization [10-12]. Solid state oxidation or creation of chemical oxidation in soil has emerged as one of the most practical remediation strategies to degrade organic pollutants in soil media [13-15]. Chemical oxidants are contacted with the contaminated soil. This starts off chemical reactions resulting in oxidative breakdowns via electron transfer processes or generation of free radical species. Hydrogen peroxide, persulfate, ozone, calcium peroxide, and permanganate are deployed as common oxidants (Table 1). The chemical activation methods are generally activated by metals, change in soil pH, or heat that creates free radical oxidants that further oxidize the organic pollutant. The sensitivity of treatment to matrix conditions will vary with different oxidants and types of contaminants [16] (Table 2). In practice, many of these remedial techniques require additional activation methods. However, they have drawbacks, including high costs, toxicity to organisms, and detrimental effects on soil properties.
The remediation of hydrocarbon or other organic contaminants in soil and water with stabilized chlorine dioxide (ClO2) represents an attractive solution. Due to its reaction selectivity and a strong chemical oxidizing agent, CLO2 has been reported to be an efficient oxidant of organic pollutants [17]. ClO2 is commonly referred to as a paramagnetic radical that is safe, economic and environmentally friendly to use in remediation applications. Contrary to many chemical oxidation techniques, the ClO2 oxidation systems does not require changing the soil pH to achieve a high degree of oxidation level. As a strong, water-soluble oxidant, it remains effective at a broad pH-range from 4-11 and does not hydrolyze in aqueous solution [18]. Furthermore, a paramount advantage of CLO2 is that it does not react with the treated system matrix to form halogenated by products compared to using free chlorine as an oxidant.
The reaction mechanism of organic pollutant by ClO2 has been investigated [19,20]. ClO2 reacts with a high degree of oxidation with the pollutant by attacking the atoms with the highest electron density. The pollutant loses electron and produces reactive intermediate. Subsequently, the unstable intermediate product participates in the ensuing reaction by undergoing molecular rearrangement and binding to ClO2 or itself, which results in the formation of oxidation products.
At present, the evaluation of efficacy of ClO2 composite powder as a chemical oxidant for remediating petroleum hydrocarbon in soils has not been explored. Thus, the main purpose of this study is to examine the oxidative degradation capacity of ClO2 and the resulting effects on soil chemistry.
Material and Methods
Measurement of soil moisture
The measurement of the soil moisture was determined by the gravimetric method. A representative soil subsample was collected, weighed before drying at 105oC ± 2 for 72 hours and then reweighed. The percent soil water is given according to Equation 1:
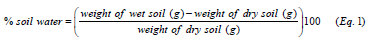
Dutridy-10 when mixed into the moist soil is expected to generate ClO2 radicals. In order for ClO2 to be active under the current investigation, it must be able to absorb moisture present in the soil to yield the following reaction: 5NaClO2 +4 HCl → 4 ClO2 + 2 H2O + 5 NaCl.
Chemical
Dutridy-10 is a composite powder that contains 10% ClO2, moisture absorbing powder, sodium chlorite and natural silicates. The product is manufactured by Duka Production LTD. To carry out the study, 400 g of contaminated soil was weighed and treated at room temperature with Dutridy containing 10% ClO2 as the source of ClO2 and tap water as well. The mixture was then homogenized mechanically in a 72 Oz Ninja Blender for 15 seconds to simulate the Micro-EnfractionaterTM. The treated matrix was then transferred into a 500mL glass, labelled with a permanent marker, and its temperature measured with a laser thermometer and recorded as a result of heat released by the exothermic reaction. The glass jar was sealed and the system was allowed to react overnight in the dark. The treated samples were subsequently delivered to a licensed laboratory in Calgary for analysis.
Soil samples collection and analyses
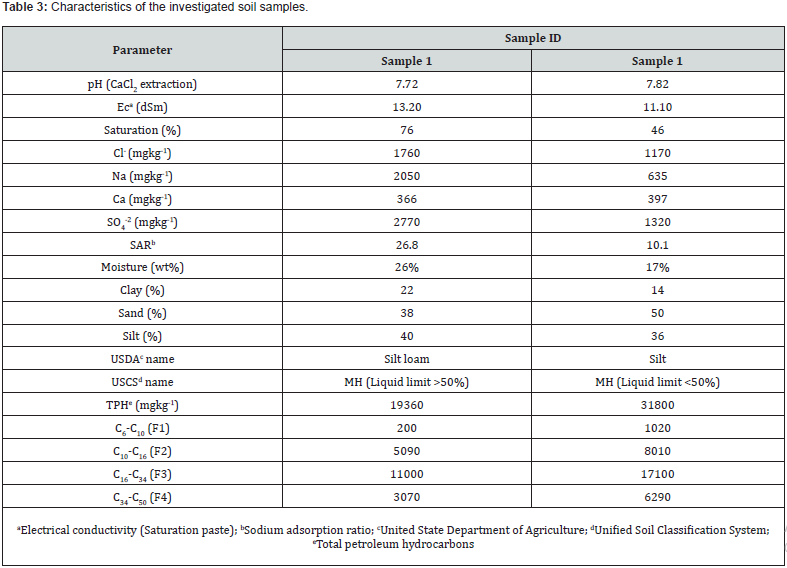
The two soils samples impacted with petroleum hydrocarbon were collected from two different sites located in northern Alberta. The samples were transferred into 20L plastic pails, labelled, mechanically homogenized, sealed and then transported to Calgary where they were stored in a fridge at 6oC. The hydrocarbon analyses were conducted by a licensed laboratory located in Calgary. Additional soil physical and chemical properties were conducted internally [21] and are summarized in Table 3. Quality assurance and quality control were maintained throughout the study and conducted as specified by manufacturer. All the internal analyses were conducted in duplicate with the average value reported.
Carbonates screening test
The hydrocarbon contaminated soil matrices were screened for the presence of carbonates. The test was performed using 0.1N and 1N HCl, respectively. Samples of the contaminated soil material were placed in a Pyrex spot plate to which 1mL of each HCl solution was added. The extent of effervescence was visually determined as an indication for presence of carbonates. The qualitative test was carried out in triplicate.
Experimental Design
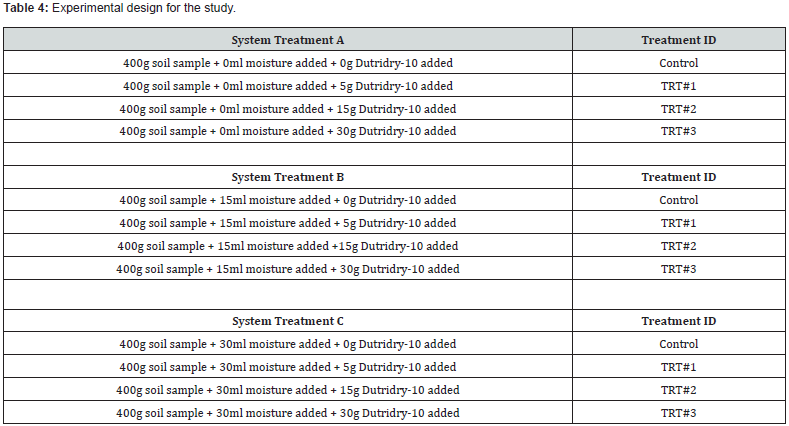
The experimental design for treatment with Dutridry-10 involving the two soil samples is depicted in Table 4. The set up consisted of three distinct systems with four treatments within a given system. Inorganic analyses and moisture determination were conducted in duplicate, and the average reported.
Results and Discussion
Soil moisture
Moisture content in the samples were 26% in sample 1 and 17% in sample 2, respectively. In sample 2, this value represents 104g of initial water content in the matrix and in sample 2 matrix, it was calculated to be 68g of initial water content. Therefore, Dutridy-10 when incorporated in the contaminated soil matrices was able to absorb the soil moisture to activate the formation of ClO2. An exothermic reaction was produced by the reaction of the oxidant with the soil matrix and measured with a laser thermometer. Heat produced increased with incremental treatment with Dutridy-10. In the experimental systems delta temperature in TRT#1 was on average 3oC while in TRT#2 and TRT#3, it was 5 and 14oC, respectively.
Carbonate mineral screening test
The intensity of the effervescence reactions in presence of HCl is an indication of the level of carbonate minerals such as calcium carbonate (CaCO3) or magnesium carbonate (MgCO3) or other types of carbonate minerals in the soil samples. Carbonate minerals do not appear to be present in significant concentration level in either of the hydrocarbon contaminated soil matrix. At 0.10 N HCl, no effervescence reaction was denoted in the samples. The 1N HCl yielded a very weak fizzing reaction in both samples.
Chemical oxidation of hydrocarbon by ClO2
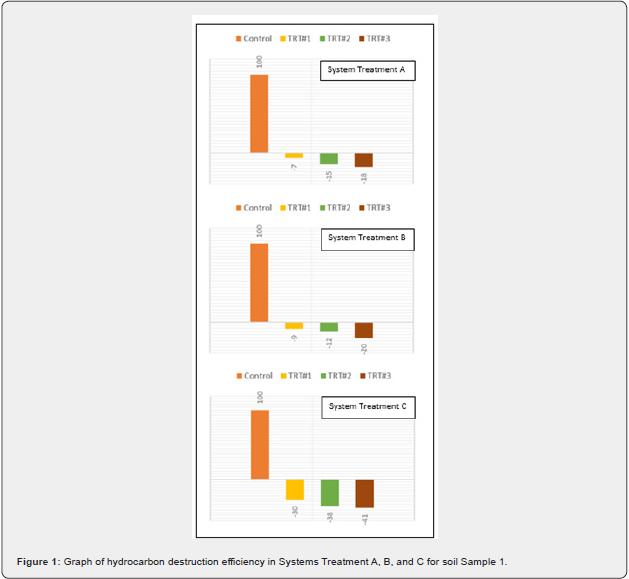
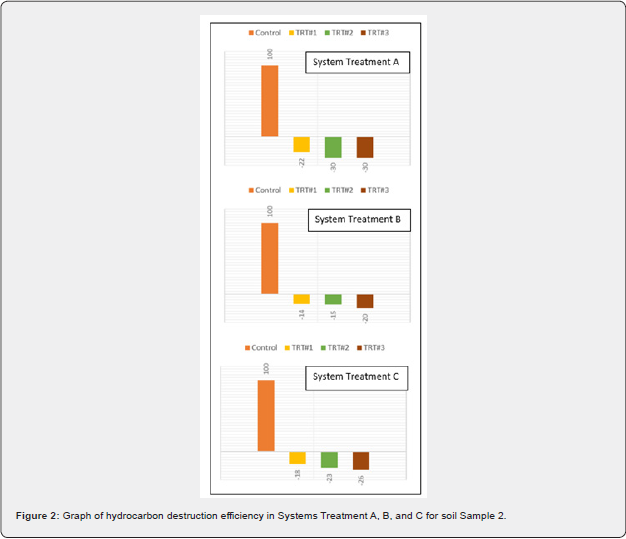
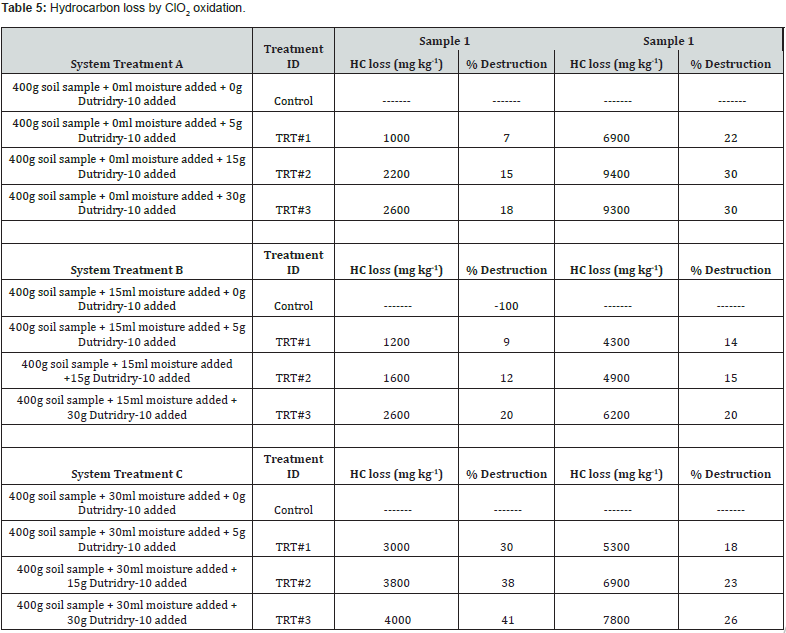
Results of petroleum hydrocarbon destruction by ClO2 is depicted in Table 5. Average TPH concentration in sample 1 was 19,360 while in sample 2, it corresponded to 31,800mg/kg. Destruction efficiency on a percent basis (%DE) of hydrocarbon in a treatment was determined by Equation 2:

Across the experiments, heat, pH, and soluble salts were parameters also followed. The pH values fractional decreased with increasing moisture and ClO2. EC values increased linearly across all treatments with higher levels of Dutridry-10. The reaction of CLO2 with the contaminated matrices produced an exothermic reaction. Average delta temperature was +3oC in systems treated with 5g of Dutridry-10. For systems treated with 15g of Dutridry-10, the average delta temperature was +5oC while an average delta temperature of +14oC was denoted in all systems treated with 30g of Dutridry-10. ClO2 radical formation is activated when the substance contacts with the soil moisture. As a strong oxidizing reagent, CLO2 transformed the petroleum hydrocarbon compounds into less harmful intermediates or end-products such as CO2. Although complete mineralization is the primary objective, partial chemical oxidation by CLO2 radical resulting in long-chain of the petroleum hydrocarbon compounds converted into lesscomplex, more dissolvable, and easily biodegradable substances or intermediates should be regarded also as a beneficial outcome. A comprehensive analysis of the % destruction efficiency trend is depicted in Figure 1 & 2, for the respective soil samples. Destruction efficiency ranged from 7 to 41% in sample 1 and from 14 to 30% in sample 2. The effectiveness of ClO2 radicals at transforming the soil hydrocarbon contaminants in sample 1 increases with increasing dosage of Dutridry-10 and soil moisture of the system. However, it was observed in sample 2 that contaminant degradation efficiency by ClO2 was rather higher at low soil moisture and increased Dutridry-10 dosage. According to this study, 5 g of Dutridry-10 combined with a soil mass water (i.e., 104g + 30g = 134g H2O) ratio 3:1 is the most appropriate combination to avoid using more water and achieving economical dosage of Dutridry-10 for treating sample 1. On the other hand, sample 2 is represented by a soil mass water (i.e., 68g + 0g = 68g H2O) ratio 6:1 with a dosage of 5g of Dutridry-10.
Hydrocarbon constituents’ interactions with soils are specific, dynamic, can be reversible or irreversible, and affected by soil solution tension and chemistry, organic matter, and types of soil mineralogy [22,23]. Taking into account the higher sand content as well as the soil mass water rater ratio, and higher F1 and F2 fractions content in sample 2 relative to sample 1, may have contributed to more hydrocarbon mass transfer into the soil solution phase. The dissolved constituents are characterized by labile bonds that can readily be attacked and broken by the ClO2 radicals in the solution. All fractions seemed to have been degraded within each soil sample. In relative term of concentration, increasing trend in degradation in both samples was F1 ≈ F2 ≈ F3 > F4 as indicated by the chromatograms data and higher ClO2 concentration resulted in higher degradation. This clearly illustrates the non-specific nature of ClO2 at degrading various organic substances. Similar results have been reported elsewhere [24]. The degradation mechanism of the hydrocarbon constituents may be occurring in two stages. The first fast-reaction stage involves initial electron transfer by Equation 3:
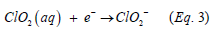
Correspondingly, residual ClO2 and the generated hypochlorous acid (HOCl) partially dissociates, forming hypochlorite anion, ClO – and would recurrently react with the contaminants. The process is illustrated through Equation 4 as determined by the relative remaining reaction time.
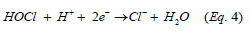
Chlorine dioxide radicals can also cause non-ionized hydrolysis of water, Equation 5, which gives rise to the formation of •OH in the CLO2 oxidation system [28].
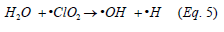
ClO2 has been substantiated as one of the most auspicious
oxidants and disinfectants. It has more oxidative capability and
disinfection power than chlorine and able to react with a variety of
organic substances. The most common and simplest components
of petroleum hydrocarbons are alkanes with formula CnH2n+2. C-C
bonds are non-polar and C-H bonds are also relatively non-polar.
ClO2 reactions with organic compounds almost does not produce
hazardous byproducts such as trihalogenomethanes. The possible
pathway for the degradation of the organic compounds by ClO2 is
mainly electrophilic. Essentially, ClO2 reacts with the hydrocarbon
constituents as a pure oxidant functioning primarily as a oneelectron
acceptor [29]. In practical term, the reaction from the
formation of the C=O and O-H bonds is exothermic and generates
CO2 and H2O. Many studies have reported that the degradation
of organic compounds by ClO2 were not pH dependent within
the range 4.5-9.5 [25] in which both soil samples fall. However,
ionizable organic compounds degradation by ClO2 are generally
pH-dependent while pH has no effect on non-ionizable organic
compounds [26,27]. Since most of the hydrocarbon constituents
in both investigated soil samples are non-ionizable organic
compounds and non-polar, one should expect that the degradation
process to be pH independent. The effect of the various
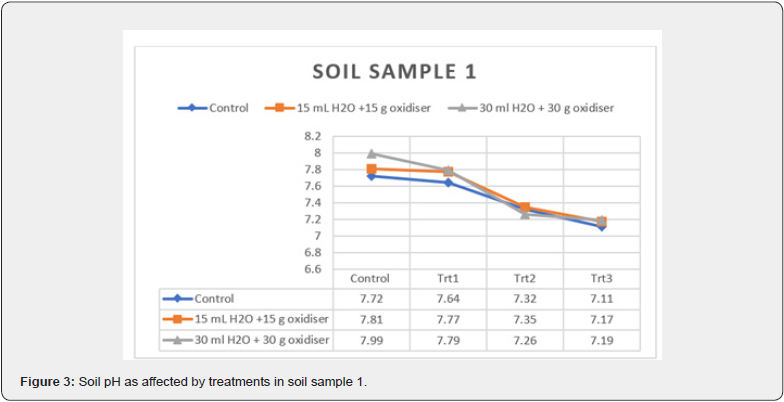
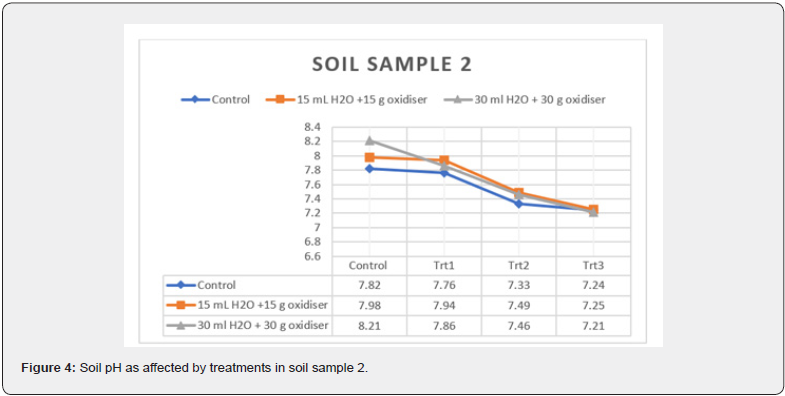
treatments on soil pH of the soil samples are plotted in Figure 3
& 4, respectively. There has been a decrease in the soil pH in all
treated systems. The delta changes in the soil slurries (i.e., initial
soil pH-final soil pH) increase with increasing ClO2 concentration
and moisture addition. Additionally, delta changes in soil pH in
sample 1 ranged in decrease from 0.08 to 0.80 while in sample
2, it was from 0.04 to 1.01. The results imply the formation of
HOCl as a secondary oxidant during the oxidation of ClO2, which
could cause a decrease in the soil pH in both samples. It was
also denoted a greater change in soil sample 2 pH in contrast to
soil sample 1. This difference could be ascribed to greater HOCl
formation in soil sample 2, which could not be offset by the lower soil buffering capacity. There was no contribution to soil salinity
of the soil samples by the various ClO2 treatments. The effects
of the ClO2 various treatments on the soil samples salinity were
analyzed. Sodium adsorption ratio (SAR) affects water intakes of
soil and is given by the formula: [Na] (([Ca] + [Mg])/2)1/2, where
all concentrations are expressed as milliequivalents of charge
per liter is an indication of the amount of Na relative to Ca and
Mg in the soil solution. SAR values within and across treatments
for each soil sample remained relatively unchanged relative to
the controls. Soil electrical conductivity (EC), a measure of the
amount of salts, increases equally in both samples as CLO2 dosage
increased. The biggest contribution to increase in the soil EC
values was attributed to higher levels of Cl- and 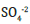 . Oxidative
transformations of sulfur containing compounds by ClO2 has
been documented [30]. As a direct effect of ClO2, concentration
of
. Oxidative
transformations of sulfur containing compounds by ClO2 has
been documented [30]. As a direct effect of ClO2, concentration
of 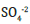 increases in the soil samples with increasing ClO2 dosage.
Increase in
increases in the soil samples with increasing ClO2 dosage.
Increase in 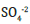 level in sample 1 ranged from 54% to 100% while
in sample 2, it was from 90% to 100%. As a direct benefit,
level in sample 1 ranged from 54% to 100% while
in sample 2, it was from 90% to 100%. As a direct benefit, 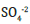 can
serve as a source of nutrient fertilizer in the soil for plants during
reclamation.
can
serve as a source of nutrient fertilizer in the soil for plants during
reclamation.
Cost analysis
Dutridry-10 is typically sold in bulk for $10/kg. At an effective application rate of 2.5g: 400g of hydrocarbon contaminated soil (i.e., 6.25g: 1kg of hydrocarbon contaminated soil), the cost of the product will be $0.0625/kg per 2000 ppm hydrocarbon oxidized. The typical hydrocarbon concentration a contaminated soil matrix is 120,000mg/kg. Therefore, for a target reduction (TR) of hydrocarbon concentration to 20,000mg/kg in the post treated soil matrix entailed a reduction, Equation 6:

Hence, we obtain 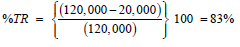
Furthermore, the ensuing calculation was also performed to determine the amount of dutridry-10 product (Kg) that should be applied to achieve the above 83% reduction assuming 1kg of hydrocarbon contaminated soil, Equation 7:
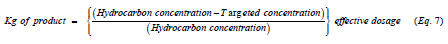
and yields
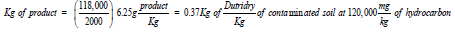
Strategically, the product could be easily incorporated mechanically with on-site equipment at no additional operational cost to the remedial program. The author believed that mixing Dutridry with the Micro-EnfractionatorTM would have the potential to increase the efficiency of the oxidizer by as much as 20-35% through improved contact surface area for reaction. Hence, an overall reduction in treatment cost.
Conclusion
It appears that ClO2 has the potential of oxidizing hydrocarbon in contaminated soils. The effectiveness of ClO2 radicals at transforming the soil hydrocarbon contaminants increases with increasing dosage and soil moisture of the system. However, the most economical dosage was a ratio of 1g product:27 g of contaminated soil. Destruction efficiency ranged from 7 to 41 % in sample 1 and from 14 to 30% in sample 2. Increasing trend in hydrocarbon degradation in both samples was F1 F2 F3 > F4. There has been a decrease in the soil pH in all treated systems. The delta changes in the soil pH slurries increase with increasing ClO2 concentration and moisture addition. The results imply the formation of HOCl as a secondary oxidant during the oxidation of ClO2, which could cause a decrease in the soil pH in both samples. Soil buffering capacity will influence the extent of soil pH lowering.
Acknowledgment
The authors are thankful for the support provided by Encana Corporation.
Conflict of Interest
The authors acknowledge that the preparation of this paper was free of any conflict of interest.
dropdown Start here
References
- Saint-Fort R (2016) Ex situ surfactant-enhanced bioremediation of NAPL-impacted vadose zone. In: Larramendy ML, Sonia S (Eds.), Soil Contamination: Current Consequences and Further Solutions. INTECH Publisher, pp. 307-327.
- Egbe E, Thompson D (2010) Environmental challenges of oil spillage for families in oil producing communities of the Niger Delta region. JHER 13: 24-34.
- Effiong SA, Etowa UE (2012) Oil spillage cost, gas flaring cost and life expectancy rate of the Niger Delta people of Nigeria. Advances in Management & Applied Economics 2(2): 211-228.
- Huan G, Manli W, Heng L, Xu Y, Liu Z (2022) Effect of petroleum hydrocarbon pollution levels on the soil microecosystem and ecological function. Environmental Pollution 293: 1-10.
- Chikere CB, Okpokwasili GC, Chikere BO (2011) Monitoring of microbial hydrocarbon remediation in the soil. Biotech 1(3): 117-138.
- Truskewycz A, Gundry TD, Khudur LS, Kolobaric A, Taha M, et al. (2019) Petroleum Hydrocarbon contamination in terrestrial ecosystems-fate and microbial responses. Molecules 24(18): 3400-3421.
- Adam G, Duncan HJ (1999) Effect of diesel fuel on growth of selected plant species. Environ Geochem Health 21: 353-357
- Hassanshahian M, Cappello S (2013) Crude oil biodegradation in marine environments. In: Rolando C (Ed.), Biodegradation-Engineering Technology. INTECH Publisher.
- Akpoghelie JO, Ugbuku UA, Esemedafe UJ (2021) A review of oil spill pollution and air quality in the Niger Delta: Causes, Effects, and Control. J Chem Soc Nigeria 46(5): 849-858.
- Wentzel A, Ellingsen TE, Kotlar HK, Zotchev SB, Holst MT (2007) Bacterial metabolism of long-chain n-alkanes. Appl Microbiol Biotechnol 76(6): 1209-1221.
- Fine P, Graber ER, Yaron B (1997) Soil interactions with petroleum hydrocarbons: Abiotic processes. Soil Technology 10(2): 133-153.
- Wang L, Cheng Y, Naidu R, Bowman M (2021) The key factors for the fate and transport of petroleum hydrocarbons in soil with related in/ex situ measurements: An overview. Frontiers in Environmental Science 9: 1-15.
- Apul OG, Dahlen P, Delgado AG, Sharif F, Westerhoff P (2016) Treatment of heavy, long-chain petroleum hydrocarbon impacted soils using chemical oxidation. J Environ Eng 42(12): 04016065.
- Collivigneralli MC, Vaccari M, Abba A, Canato M, Sorlini S (2018) Wet oxidation of fine soil contaminated with petroleum hydrocarbons: A way towards a Remediation Cycle. Environments 5(69): 1-13.
- Daâssi D, Almaghribi FQ (2022) Petroleum-contaminated soil: environmental occurrence and remediation strategies. Biotech 12(6): 139.
- Huling SG, Pivetz BE (2006) In-situ chemical oxidation (N0. EPA/600/R-06/072). Environmental Protection Agency, Washington, DC. Office of Water.
- Sun Y, Niu WK, Hu XJ, Ma XH, Sun YJ, et al. (2020) Oxidative degradation of polycyclic aromatic hydrocarbons in contaminated industrial soil using chlorine dioxide. Chem Eng J 394: 124857.
- Stevens AA (1982) Reaction products of chlorine dioxide. Environ Health Perspect 46: 101-110.
- Nie S, Lin X, Wu Z, Zhan L, Yin G, et al. (2014) Kinetic studies of oxidation of the lignin model compounds by chlorine dioxide. Chem Eng J 241: 410-417.
- Jia XH, Feng I, Liu YZ, Zhang LQ (2017) Oxidation of antipyrine by chlorine dioxide: Reaction kinetics and degradation pathway. Chem Eng J 309: 646-654.
- Klute A (1996) Chemical and microbial properties. In: Methods of Soil Analysis. (2nd edn), American Society of Agronomy: Madison, WI, pp. 167-693.
- Bhuyan SJ, Latin MR (2012) BTEX remediation under challenging site conditions using in-situ ozone injection and soil vapour extraction technologies: a case study. Soil Sediment Contam 21(4): 545-556.
- Yeung AT, Gu YY (2011) A review on techniques to enhance electrochemical remediation of contaminated soil. J Hazard Mater 195: 11-29.
- Ma YL, Gu N, Gao KT, et al. (2016) Remediation of anthracene-contaminated soil by ClO2 in the presence of magnetic Fe3O4-Cu@montmorillonite as catalyst.
- Rav-Achach, Blits R (1985) The different reaction mechanisms by which chlorine and chlorine dioxide react with polycyclic aromatic hydrocarbons (PAHs) in water. Water Res 19(10): 1273-1281.
- Ben WW, Shi YW, Li WW, Zhang Y, Qiang Z (2017) Oxidation of sulfonamide antibiotics by chlorine dioxide in water: kinetics and reaction pathways. Chem Eng J 327: 743-750.
- Napolitano MJ, Stewart DJ, Margerum DW (2006) Chlorine dioxide oxidation of guanosine 5’-monophosphate. Chem Res Toxicol 19(11): 1451-1458.
- Macron J, Mortha G, Marlin N, Molton F, Duboc C, et al. (2017) New insights into the decomposition mechanism of chloride dioxide at alkaline pH. Holforschung 71: 599-610.
- Liu JQ, Huang JL, Su XY, Xiangyu C, Ying J (2006) Degradation of anthracene, pyrene, benzo(a)-anthracene in aqueous solution by chlorine dioxide. Sci in China Series B: Chemistry 49: 565-572.
- Kutchin AV, Rubtsova SV, Lezina OM, Sudarikov DV, Frolova LL, et al. (2017) Studies on oxidative transformations of thiols, sulfides and alcohol in the presence of chlorine dioxide. Pure Appl Chem 89: 1379-1401.






























