Abstract
Background: Post-thoracotomy pain is a significant concern; effective analgesia is crucial for recovery. Thoracic epidural analgesia (TEA) is widely regarded as the reference technique following open thoracotomy; however, if TEA is contraindicated, catheter-based wound infiltration (CBWI)
emerges as a viable alternative.
Methods: Retrospective, single-center cohort study of consecutive adults who underwent elective thoracotomy. Intensity of pain was measured
using a verbal numerical rating scale (VRS; 0-10) at rest and during coughing/deep breathing at predetermined time points: t0 (ICU arrival), t1
(6 hours), t2 (12 hours), t3 (24 hours), t4 (48 hours), and t5 (72 hours). Analyses employed linear mixed-effects models (group, time, random
intercept for subject), adjusted for age, sex, ASA, surgical approach, anticoagulation therapy, chronic pain.
Results: Data set included 81 patients (28 female, 53 male), of these 41 received TEA, 40 CBWI. In CBWI group, conversion from video-assisted to
open thoracotomy occurred in 16 cases. Across t0-t5, NRS trajectories exhibited substantial similarity between groups with small between‑group
disparities at discrete time points. Systemic co‑analgesic use was higher in CBWI, while serious adverse events were uncommon in both groups.
Duration of catheter use was comparable (PDC: 92±27h, CBWI: 83±59h), although total ropivacaine doses differed (TEA: 1067±468mg, CBWI:
878±182mg).
Conclusions: In this real‑world cohort, CBWI emerged as a feasible alternative when TEA was contraindicated or declined. Given the observational
design and potential confounding by indication, estimates should be interpreted with caution and in the context of randomized evidence that
favors TEA over subpleural/intercostal approaches after thoracotomy.
Keywords:Catheter-Based Wound Infiltration; Peridural Anesthesia; Postoperative Pain Management; Ropivacaine; Sufentanil; Thoracotomy
Abbreviations:TEA: Thoracic Epidural Analgesia; VATS: Video-Assisted Thoracoscopic Surgeries; ERAS: Enhanced Recovery After Surgery; PDC: Peridural Catheter; CBWI: Catheter-Based Wound Infiltration; CPSP: Chronic Postsurgical Pain Preliminary results of the study have been presented as an electronic poster presentation at the DGAI Annual Meeting 2024, September 10th. - 14th., Kassel, Germany (DOI:10.13140/ RG.2.2.21433.38246) and as oral presentation at the 2024 DGT Annual Meeting. Zentralbl Chir 149 (2024), S01, S99-S100 (DOI 10.1055/s-0044-1788120)
Introduction
In thoracic surgery, thoracotomy remains an essential approach despite the growing adoption of minimally invasive techniques, including video- or robot-assisted thoracoscopic surgery. However, thoracotomy is among the most painful surgical procedures, comparable to laparotomy and mastectomy [1]. Contributors to this high pain burden include rib retraction or resection, rib fractures, intercostal nerve injuries, and pleural irritation from chest tubes. The absence of postoperative epidural analgesia, female sex, preexisting opioid therapy, and chronic pain are among the strongest risk factors for higher postoperative pain intensity. In addition, antidepressant use has been identified as an independent risk factor for elevated pain levels [2]. Effective analgesia, together with preoperative patient education, improves patient satisfaction and is a key prerequisite for implementing fast-track or enhanced recovery after surgery (ERAS) pathways [2-6].
Inadequate analgesia after thoracic surgery is associated with an increased risk of postoperative complications and may exacerbate existing comorbidities. In patients with coronary artery disease, which is common in thoracic surgical populations, insufficient pain control increases the risk of coronary complications [1]. Moreover, thoracic pain can cause splinting with reduced inspiratory effort and impaired cough, thereby promoting postoperative pulmonary complications [7]. Chronic postsurgical pain is common and can substantially impair quality of life. A high incidence of chronic pain has been reported after both thoracotomy and thoracoscopy, with rates varying by the analgesic regimen used [7-12].
These considerations underscore the importance of effective pain management as a primary goal of perioperative care. Regional anesthesia techniques, particularly thoracic epidural analgesia (TEA), are considered the gold standard for post-thoracotomy pain control. However, TEA is not feasible for all patients [13]. Contraindications include patient refusal, anatomic or pathophysiological limitations, and perioperative anticoagulation. In addition, more than 10% of planned videoassisted thoracoscopic surgeries (VATS) require conversion to open thoracotomy, which can make preoperative placement of an epidural catheter (peridural catheter [PDC]) impractical [14,15]. TEA also requires an anesthesiology-led acute pain service [16], which increases resource use and costs.
As an alternative, an extrapleural catheter placed intraoperatively at the surgical site enables continuous catheterbased wound infiltration (CBWI) with local anesthetics [17,18]. This approach has been proposed as a cost-effective and clinically viable substitute for TEA; however, further studies are needed to clarify its role in thoracic surgery analgesia [17,19-22]. This clinical study evaluates continuous postoperative pain management strategies in thoracic surgery. Specifically, it compares TEA with wound infiltration analgesia delivered via an intercostal catheter connected to an elastomeric pump that continuously administers a fixed dose of local anesthetic.
We did not test a noninferiority hypothesis; instead, we estimated covariate-adjusted between-group differences over time and interpreted them descriptively given potential residual confounding. The null hypothesis (H0) was that catheter-based infiltration analgesia does not differ from TEA with respect to postoperative pain intensity. The alternative hypothesis (H1) was that postoperative pain intensity differs between the two pain management strategies during the postoperative period. The primary outcome was postoperative pain intensity, assessed using a verbal numeric rating scale (VRS; 0-10) at rest and during coughing/deep breathing at pre-specified time points postoperatively.
Patients and Methods
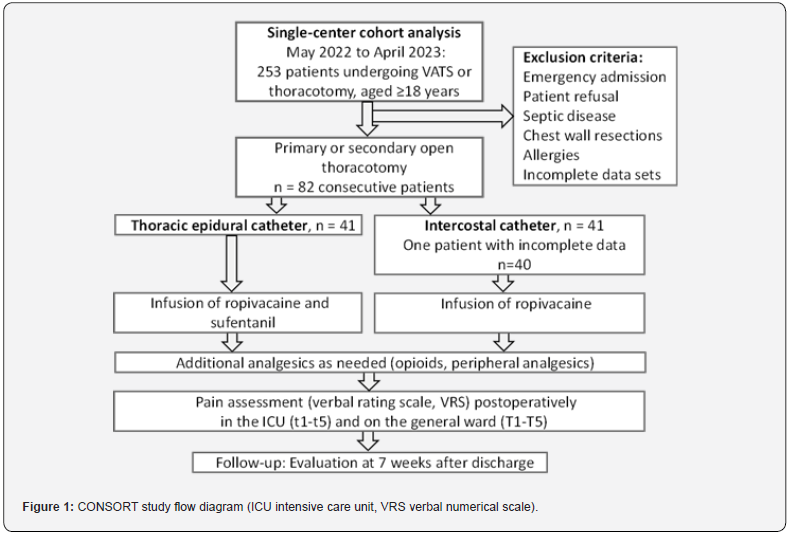
We conducted a retrospective, single-center cohort study at the Department of Cardiac and Thoracic Surgery, Otto-von-Guericke- University Magdeburg (Saxony-Anhalt, Germany), including consecutive adult patients undergoing elective thoracotomy (Figure 1). Study design and reporting: Retrospective, singlecenter cohort of consecutive thoracotomy patients; reporting followed the STROBE statement. Outcomes and time points (harmonized): The primary outcome was postoperative pain intensity on the 0-10 numeric rating scale (VRS), assessed at rest and during coughing/deep breathing. Time points were t0 (ICU arrival), t1 (6 h), t2 (12 h), t3 (24 h), t4 (48 h), and t5 (72 h). Measurements obtained within ±2 h were assigned to the nearest prespecified time point.
Ethical Considerations
The retrospective data analysis was approved by the Ethics Committee of Otto von Guericke University Magdeburg, Germany, based on the ethical approval for querying and analyzing perioperative patient data from the “Multicenter Registry for the Collection of Pseudonymized Patient Data in the Context of Thoracic Surgery” (Thorax Registry, https://www.thoraxregister. de/), which was granted by the Ethics Committee of Witten/ Herdecke University, Germany (64/2014).
The study was conducted in accordance with the STROBE guidelines [23], the principles of the Declaration of Helsinki [24,25], and the data protection regulations of the state of Saxony- Anhalt, Germany. Anesthetic and thoracic surgical care adhered to established clinical standards. Exclusion criteria were acquired or congenital coagulopathy, sepsis, shock, severe hypovolemia, elevated intracranial pressure, meningitis, encephalitis, or specific neurologic disorders without prior documentation. Patients lacking required preoperative testing (spirometry or arterialized capillary blood gas analysis on room were also excluded. Additional exclusion criteria included infection at the puncture site, allergy to the local anesthetics used, selected cardiovascular conditions (severe aortic valve stenosis, intracardiac shunt, or pulmonary hypertension), decompensated liver disease, pregnancy, or female patients without a documented negative pregnancy test.
Intraoperative Management
Before surgery, all patients underwent a standardized preanesthesia assessment, including counseling and physical examination by an anesthesiologist. Prior to induction, a peripheral intravenous catheter and an arterial line were placed under local anesthesia (2 mL lidocaine 1%). Continuous monitoring included pulse oximetry and electrocardiography. General anesthesia was induced in a weight-adjusted manner with sufentanil (15-30μg; 0.1-0.3 μg·kg⁻¹), propofol (1.0-1.5 mg·kg⁻¹), and rocuronium (0.6- 0.9 mg·kg⁻¹). Intraoperative fluid therapy consisted of crystalloids. Prophylactic antibiotics included a single dose of cefuroxime 1.5g, and dexamethasone 0.1mg·kg⁻¹ was administered for postoperative nausea and vomiting prophylaxis. After orotracheal intubation with a double-lumen endotracheal tube, pressurecontrolled mandatory ventilation was initiated with a tidal volume of 6-8mL·kg⁻¹, FiO₂ 0.6, and PEEP 6 cmH₂O. Respiratory rate was adjusted to maintain normocapnia (PaCO₂ approximately 40-45 mmHg). A central venous catheter was placed in the ipsilateral internal jugular vein. Urine output and body temperature were continuously monitored via a urinary catheter.
To maintain anesthesia, inhalation of desflurane or sevoflurane (~ 0.3 - 0.6 MAC) or continuous infusions of propofol 2 % (4 mg·kg- 1·h⁻¹) were used as well as remifentanil (0.2-0.3 μg·kg⁻¹·min⁻¹) or repeated bolus injections of sufentanil (10 - 15 μg) as required, which were continued until extubation. After thoracic surgery, all patients were transferred to a postoperative monitoring unit (ICU) for at least 24 hours of intensive observation. Postoperative monitoring included continuous assessments of vital parameters and pain evaluation at defined intervals every 4 - 6 hours over five observation time points (t₀ upon admission, t₅ at discharge). From the first postoperative day onward, pain intensity scores were documented once daily until discharge (t₀ at admission to the general ward, t₅ at discharge).
Catheter-based Pain Protocols
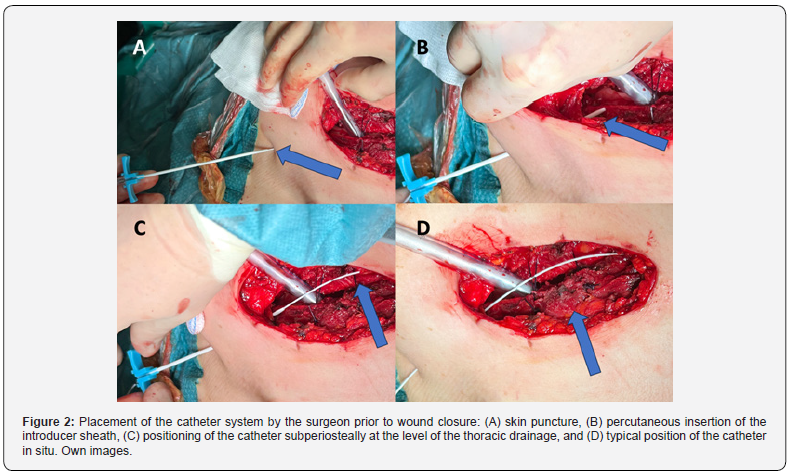
For TEA catheters infusions contained ropivacaine 0.2 % and sufentanil 1 μg/ml administered as an initial bolus of 6 ml followed by infusion rates of 6 - 8 ml/h (TEA). Wound infiltration catheters (InfiltraLong®, and FuserPump®, PAJUNK Ltd., Geisingen, Germany) were filled with ropivacaine 0.2 % at infusion rates of 6 -8 ml/h, following an intraoperative bolus of 20 ml ropivacaine 0.75 % (CBWI, (figure 2)).
Data Collection
Perioperative data were extracted from the electronic patient record of the Department of Cardiac and Thoracic Surgery at Ottovon- Guericke University Magdeburg, Germany. Additional variables were obtained from the German Thoracic Registry including procedure type and extent, emergency classification, laboratory data, comorbidities, postoperative pain records. Supplemental data were retrieved from the electronic anesthesia protocols and laboratory report forms, including demographic data (age, height, weight), risk classification (ASA status), individual risk factors (e.g., smoking, alcohol abuse), preoperative spirometry results, blood gas values (PaO₂, PaCO₂, SaO₂, oxygenation index).
Statistical Analysis
Data were analyzed using SPSS for Windows (IBM SPSS Statistics, Version 28, Armonk, NY, USA) and graphically presented using SigmaPlot, Version 15.0, (Systat Software Inc., San Jose, CA, USA). The primary outcome was defined as postoperative pain intensity, assessed in the ICU using a verbal numeric rating scale (VRS; 0-10) at rest. An a priori power analysis indicated a required sample size of at least 52 patients, assuming a two-sided study design, an effect size of 1, an α - error of 0.05, and a power (1 − β) of 0.95. Secondary outcome parameters included reintubation rate, incidence of surgical complications, duration of the patient’s stay in the intensive care unit, total hospitalization duration, 30- day mortality and patient status after seven weeks.
Categorical variables are reported as counts and percentages. Continuous variables are summarized using measures of central tendency and dispersion. Normality was assessed with the Shapiro-Wilk test, and homogeneity of variances with Levene’s test. Normally distributed variables presented as mean±SD. Non-normally distributed variables were analyzed using nonparametric tests (Mann-Whitney U or Kruskal-Wallis tests) and visualized with boxplots (median, mean, interquartile range, and 5th-95th percentiles). NRS trajectories were modelled with linear mixed‑effects models (random intercepts for participants; fixed effects for group, time, and group×time). A priori adjustment: age, sex, ASA class, surgical approach (open vs VATS conversion), anticoagulation/antiplatelet therapy, and chronic pain/neurological comorbidity. Sensitivity analyses used inverse probability of treatment weighting (propensity scores) to mitigate confounding by indication. Time-specific contrasts were adjusted for multiplicity using the Holm-Bonferroni method. Non‑inferiority testing was not performed due to the absence of a pre‑specified margin and the retrospective design. A two-sided significance level of α = 0.05 (p < 0.05) was used for all statistical tests.
Results
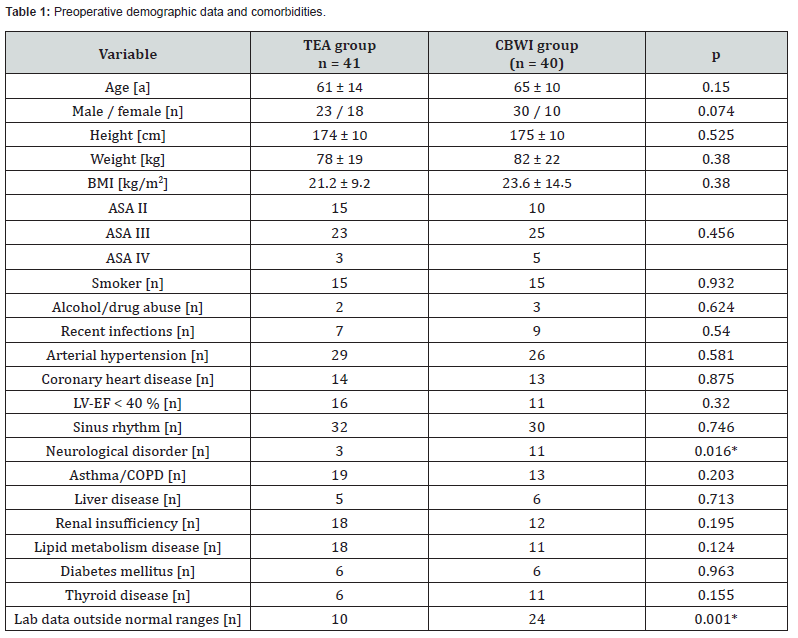
The study included 82 consecutive thoracic surgery patients: 41 in the TEA group and 40 in the catheter-based wound infiltration (CBWI) group. One patient was excluded because of incomplete data (Figure 1). Reasons for CBWI rather than a thoracic epidural catheter were conversion from VATS to open thoracotomy (n=16), refusal of epidural placement (n=11), contraindication due to ongoing anticoagulant therapy (n=10), and technical failure (n=3). Demographic, biometric, and general operative variables stratified by group are summarized in (Table 1). No statistically significant between-group differences were observed. However, neurologic comorbidities (e.g., prior stroke or polyneuropathy) and preoperative laboratory values-predominantly coagulation parameters-were more frequent/abnormal in the CBWI group.
Preoperative Characteristics
Preoperative lung function was within age-appropriate reference ranges in all patients. Overall, 11 patients were taking psychotropic medications and 13 were receiving analgesics preoperatively; distributions were similar between groups.
Surgical Indications and Intraoperative Procedures
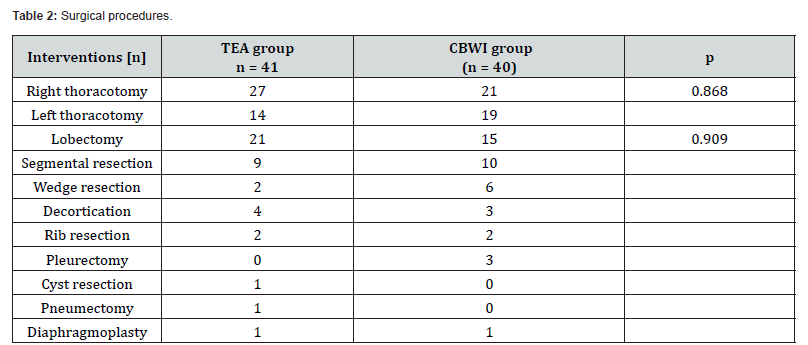
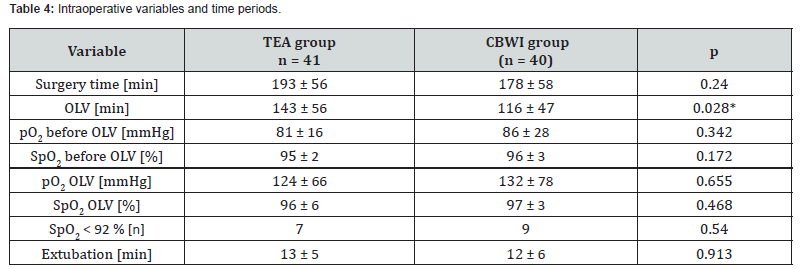
Primary surgical indications included pulmonary lesions (e.g., cysts, primary lung tumors, carcinoid tumors, or metastases), diaphragmatic repair, and pleural empyema (Table 2). Indications did not differ between groups. In total, 48 right-sided and 33 left-sided thoracotomies were performed; lobectomy (n=36) and segmentectomy (n=19) were the most common procedures. Intraoperative anesthetic use and operative variables are reported in (Tables 3, 4).
Postoperative Pain
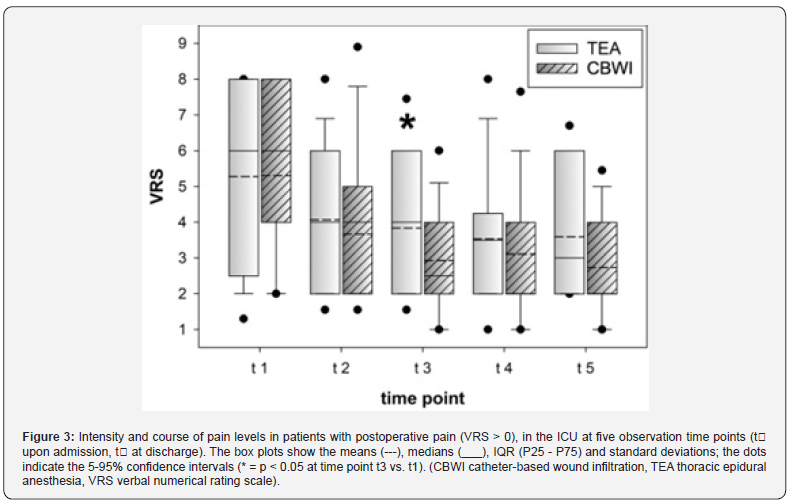
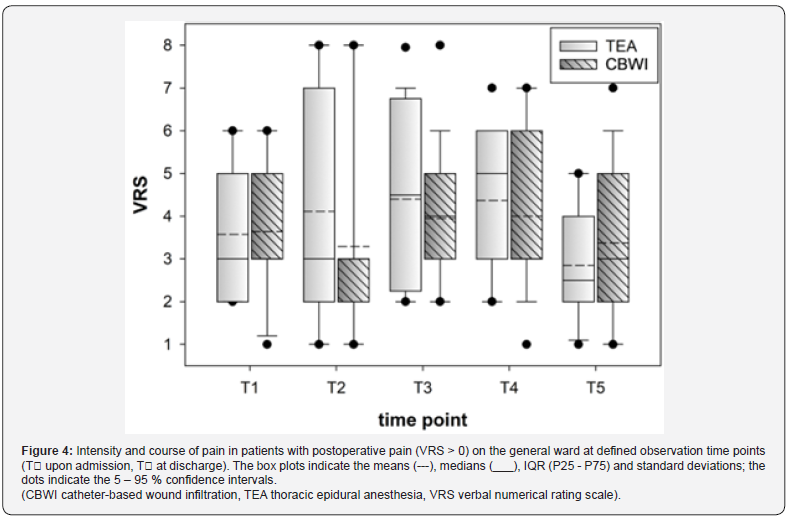
On ICU admission, 25 patients in the TEA group and 24 in the CBWI group reported pain on the VRS (0-10); five patients in each group were not yet extubated. Overall, 22 patients were pain-free (VRS=0), and the cohort median VRS score was 3.3. Pain intensity and trajectories across the ICU time points were comparable between groups (Table 5). At time point t3, patients in the TEA group reported higher pain scores (Figure 3), while more CBWI patients received metamizole; opioid dosing was similar (Table 6). After transfer to the general ward, 17 patients in the TEA group and 11 in the CBWI group reported pain (VRS>0), meaning that 53 patients were pain-free overall. The cohort median VRS score was 1; among patients reporting pain, the mean score was 3. Pain intensity and trajectories from t1 to t5 did not differ between groups (Table 7, Figure 4). At discharge, the mean pain score for the entire cohort was 2.1, with no significant between-group difference.
Additive Analgesia in ICU and Ward
In the ICU, 14 patients received metamizole, and 41 received additional opioids (Table 6). On the general ward, CBWI patients received a higher total dose of ropivacaine (712±431mg) than TEA patients (443±226mg; p<0.01). Metamizole consumption was also higher (TEA vs CBWI: 22±17g vs 31±19g; p=0.034), as was opioid consumption (TEA vs CBWI: 84±120mg vs 178±192 g; p=0.063). Epidural sufentanil reduced the required dose of local anesthetic (ropivacaine 405±334mg with sufentanil 83±87μg vs ropivacaine 518±324mg; p<0.01). Mean catheter duration was 92±27 hours in the TEA group and 83±59 hours in the CBWI group (p=0.389).
Adverse Events
Monitored adverse events included pulmonary complications, reintubation, therapy-related side effects (e.g., nausea, arterial hypotension, pruritus), delirium, tolerance of oral intake, and early postoperative mobilization. Across these outcomes (Table 8), no clinically meaningful differences were observed between groups. No severe complications occurred, including epidural hematoma, infection, or local anesthetic systemic toxicity.
Postoperative Follow-Up
Patients returned for outpatient follow-up at a mean of 43 days after discharge. Outcomes included surgical status, wound complications, paresthesia in the surgical area, and persistent pain. No major wound complication requiring reoperation occurred. Persistent pain VRS>0), typically mild to moderate, was reported by 41 patients (51%), with a median VRS score of 3 and no significant between-group difference. Postoperative analgesics included metamizole in 37 patients (46%) and long-acting opioids in five patients; postoperative analgesic use did not differ between groups.
(ASA American Society of Anesthesiologists classification, BMI body mass index, CBWI catheter-based wound infiltration, LV-EF left-ventricular ejection fraction, TEA thoracic epidural analgesia).
(CBWI catheter-based wound infiltration, TEA thoracic epidural analgesia).
(CBWI catheter-based wound infiltration, TEA thoracic epidural analgesia).
(CBWI catheter-based wound infiltration, OLV one-lung ventilation, TEA thoracic epidural analgesia).
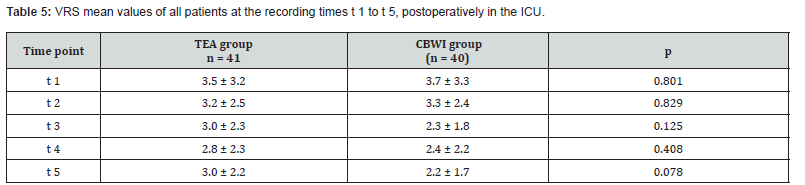
(CBWI catheter-based wound infiltration, TEA thoracic epidural analgesia, VRS verbal numerical rating scale).
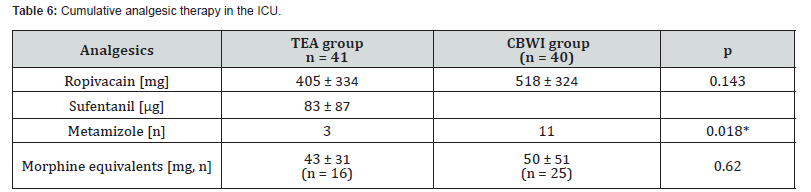
(CBWI catheter-based wound infiltration, TEA thoracic epidural analgesia).
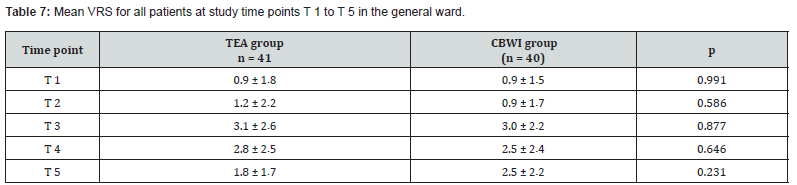
(CBWI catheter-based wound infiltration, TEA thoracic epidural analgesia, VRS verbal numerical rating scale).
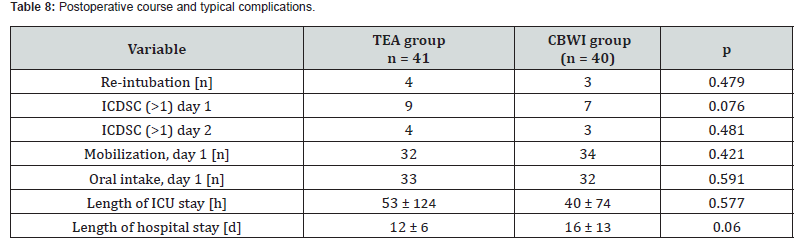
(CBWI catheter-based wound infiltration, ICDSC Intensive Care Delirium Screening Checklist, ICU intensive care unit, TEA thoracic epidural analgesia).
Discussion
In this pragmatic cohort, CBWI appeared clinically comparable to TEA with respect to early postoperative VRS trajectories, with small differences at isolated time points. These findings support CBWI as an option when TEA is contraindicated or declined. However, thoracic-specific randomized trials generally favor TEA over subpleural or intercostal approaches in terms of analgesic efficacy. Accordingly, our results should not be interpreted as evidence of noninferiority but rather as complementary realworld evidence. Thoracotomy is among the most painful surgical procedures [1]. Beyond established perioperative strategies - such as preoperative risk assessment and optimization; lung-protective ventilation (e.g. FIO₂, titration, low tidal volumes, permissive hypercapnia, appropriate PEEP, and recruitment maneuvers); as well as optimized fluid therapy [26] - postoperative pain management is central for surgical success and patient satisfaction. Regional anesthesia techniques are crucial to maximizing the potential benefits of modern thoracic surgery.
Recent research provided sufficient evidence that chronic postsurgical pain (CPSP) is a common complication after both thoracotomy and video-assisted thoracoscopic surgery [12,27]. The incidence of CPSP after thoracic surgery varies widely, with reported rates ranging from 25% and 60%. This variability is influenced by factors such as the surgical approach (minimally invasive vs. open procedures), perioperative pain management, and individual patient characteristics. For example, a study involving more than 2,300 patients undergoing VATS reported that approximately 44% developed CPSP, with 15% experiencing moderate-to-severe pain. Although thoracotomy typically results in a higher CPSP burden than VATS, both procedures carry substantial risk [27].
Several risk factors have been associated with CPSP, including younger age, female sex, preoperative pain, and postoperative complications such as wound infection. Addressing modifiable risk factors through optimized perioperative pain management is important for reducing the incidence and severity of CPSP.
The optimal approach to pain management in thoracic surgery remains debated. Effective pain control not only improves patient satisfaction but may also reduce the transition from acute to chronic pain [2]. Thoracic epidural analgesia is widely considered the gold standard after thoracotomy in many centers [6,28]; however, it is not suitable for all patients and is occasionally associated with serious complications, including rarely epidural hematoma, abscess, or direct spinal cord injury [29]. Consequently, alternative regional anesthesia techniques have gained popularity in recent years. However, due to heterogeneous evidence, a comparable consensus regarding the optimal approach in thoracic surgery has not been reached, although recent literature increasingly supports thoracic, paravertebral block, serratus anterior plane block, and erector spinae plane block [22,30].
The present clinical study compares two continuous methods for postoperative pain management: thoracic epidural analgesia using a preoperatively placed epidural catheter and wound infiltration using an intercostal catheter placed by the surgeon and connected to an elastomeric pump for continuous, fixed-dose delivery of local anesthetic [18]. The principal finding is that, in two biometrically and clinically similar groups with comparable pain levels after thoracotomy, no statistically significant differences were observed in pain intensity or pain trajectories. Given the observational design, these estimates remain susceptible to residual confounding by indication. Pain intensity, the primary outcome, was assessed postoperatively using a verbal numeric rating scale (VRS; 0-10) at rest [31].
Postoperative analgesia was generally effective, as 27% of patients reported being pain-free upon ICU admission. With a median pain score of approximately 3 across all patients, pain intensity decreased across the five ICU assessment time points (median ICU stay: 20 hours). At time point t3, patients in the CBWI group reported lower pain intensity. This finding is notable because CBWI patients received slightly higher (though not statistically significant) doses of ropivacaine, NSAIDs, and morphine equivalents but no sufentanil via the catheter. Thus, opioid-free wound infiltration with ropivacaine may provide clinically meaningful analgesia in selected patients compared with TEA using ropivacaine/sufentanil. These findings also underscore the risk of severe pain after open thoracotomy, as several patients initially reported VRS scores >5 (up to 8).
Continuous wound infiltration with local anesthetics was first introduced in general surgery for procedures such as inguinal hernia repair [32] and has also been applied to subcostal incisions, open appendectomy, and midline laparotomy [33]. The ratio of incision length to local anesthetic infusion rate appears to play an important role [34]. This technique was first investigated in thoracic surgery in 2005, with satisfactory pain control reported in 71 patients compared with TEA [17]. Comparable studies have evaluated erector spinae plane blocks versus serratus anterior plane blocks, reporting average pain scores of 4 to 5. Despite reduced surgical trauma with minimally invasive thoracoscopy, these scores are higher than those observed in the present cohort [35].
In another study, subpleural analgesia was compared with TEA using patient-controlled analgesia (PCA) [36]. TEA provided significantly better pain relief, with average VRS (0-10) scores of 2 to 3 for TEA compared with 4 to 7 for subpleural analgesia during the first 24 hours. Patients receiving TEA also required significantly less rescue medication. Although patients in that study were younger than those in our cohort, vital signs and other demographic characteristics were similar. After transfer from the ICU to the general ward, the proportion of pain-free patients increased to >65%. Median pain intensity decreased to approximately 1 but increased again after discontinuation of continuous therapy and with increased mobilization. Pain intensity and trajectories did not differ meaningfully between groups; however, the type and quantity of analgesics administered on the general ward varied, limiting late postoperative comparisons.
The study data support cohort homogeneity with respect to age, sex, biometric measures, anesthetic risk, and preoperative comorbidities. The higher prevalence of coagulation abnormalities in the CBWI group reflects allocation criteria, as coagulopathy is a contraindication to epidural catheter placement [37]. The increasing prevalence of oral anticoagulant therapy in aging populations underscores the clinical relevance of alternatives to TEA. Most secondary endpoints, including postoperative delirium incidence, reintubation rate, pulmonary complications, early mobilization, and resumption of oral intake, were similar between groups. Median hospital stay was three days longer in CBWI patients, although this difference cannot be fully explained by the variables assessed in this study. Factors such as drain management, nursing requirements, antibiotic therapy, or additional diagnostic procedures likely influenced discharge timing.
Severe complications, such as epidural bleeding, infection, or local anesthetic systemic toxicity, were not observed. Pulmonary complications, adverse effects such as nausea, arterial hypotension, or pruritus, and delirium incidence were also similar between groups. Nonetheless, limitations include the relatively short duration of catheter use and the lack of standardized continuation of pain management on the general ward. A standardized ward protocol has since been implemented, incorporating scheduled and as-needed administration of NSAIDs and WHO step III opioids. In summary, continuous pain management using intercostal CBWI catheters provided analgesia that was clinically similar to TEA in the early postoperative period while avoiding epidural-related complications such as spinal infection or hematoma. In addition, this method can be implemented readily in cases of unplanned conversion to open surgery.
Although multimodal pain strategies have advanced, clinically relevant postoperative pain persists in some patients, and chronic pain may develop in a substantial proportion, adversely affecting quality of life. Postoperative pain management in thoracic surgery should therefore remain multimodal, incorporating pharmacologic analgesia, physiotherapy, and psychological interventions when indicated. Combining regional anesthesia with systemic medications, such as NSAIDs and opioids, can improve analgesic quality in the immediate postoperative phase.
For patients who cannot receive TEA due to infection, anticoagulation, or patient preference, intercostal pain catheters represent a feasible alternative. Future research should address optimal local anesthetic concentrations and dosing, therapy duration, and the integration of adjunct analgesics into multimodal protocols. Prospective comparisons with other regional analgesic techniques, such as peripheral nerve blocks, are needed. Multicenter clinical trials tailored to different surgical approaches are critical for refining and standardizing pain management strategies in thoracic surgery. By reducing undesirable effects of systemic analgesia, improving lung function, and accelerating recovery, effective pain management may enhance patient satisfaction and improve outcomes.
Conclusions
This study suggests that continuous postoperative pain management via CBWI is clinically similar to TEA in patients undergoing thoracotomy. Both techniques achieved comparable early pain courses and overall effectiveness, as reflected by similar numeric rating scale scores at various postoperative time points, despite minor differences in supplemental analgesic consumption. Notably, CBWI represents a feasible alternative when TEA is contraindicated, such as in patients with coagulation abnormalities or those refusing epidural placement, and in scenarios of unplanned conversion from minimally invasive to open surgery. Although effective in the acute postoperative period, the persistence of mild-to-moderate chronic pain in a substantial subset emphasizes the need for comprehensive multimodal analgesia strategies. Future investigations should optimize local anesthetic dosing and catheter duration, integrate additional analgesic modalities, and directly compare CBWI with emerging regional anesthesia techniques in multicenter settings.
References
- Likar R, W Jaksch, T Aigmüller, M Brunner, T Cohnert, et al. (2017) Interdisciplinary position paper "Perioperative pain management". Schmerz 31(5): 463-482.
- Kampe S, Martin Wendland, Stefan Welter, Clemens Aigner, Thomas Hachenberg, et al. (2018) Independent Predictors for Higher Postoperative Pain Intensity During Recovery After Open Thoracic Surgery: A Retrospective Analysis in 621 Patients. Pain Med 19(8): 1667-1673.
- Piccioni F, R Ragazzi (2018) Anesthesia and analgesia: how does the role of anesthetists changes in the ERAS program for VATS lobectomy. J Vis Surg 4: 9.
- Umari M, Stefano Falini, Matteo Segat, Michele Zuliani, Marco Crisman, et al. (2018) Anesthesia and fast-track in video-assisted thoracic surgery (VATS): from evidence to practice. J Thorac Dis 10(Suppl 4): S542-S554.
- Batchelor TJP, Neil J Rasburn, Etienne Abdelnour-Berchtold, Alessandro Brunelli, Robert J Cerfolio, et al. (2019) Guidelines for enhanced recovery after lung surgery: recommendations of the Enhanced Recovery After Surgery (ERAS(R)) Society and the European Society of Thoracic Surgeons (ESTS). Eur J Cardiothorac Surg 55(1): 91-115.
- Loop T (2016) Fast track in thoracic surgery and anaesthesia: update of concepts. Curr Opin Anaesthesiol 29(1): 20-25.
- Muehling BM, Gisela L Halter, Hubert Schelzig, Rainer Meierhenrich, Peter Steffen, et al. (2008) Reduction of postoperative pulmonary complications after lung surgery using a fast-track clinical pathway. Eur J Cardiothorac Surg 34(1): 174-180.
- Gottschalk A, EA Ochroch (2008) Clinical and demographic characteristics of patients with chronic pain after major thoracotomy. Clin J Pain 24(8): 708-716.
- Wildgaard K, J Ravn, H Kehlet (2009) Chronic post-thoracotomy pain: a critical review of pathogenic mechanisms and strategies for prevention. Eur J Cardiothorac Surg 36(1): 170-180.
- Takenaka S, Ayano Saeki, Norihiko Sukenaga, Ryusuke Ueki, Nobutaka Kariya, et al. (2020) Acute and chronic neuropathic pain profiles after video-assisted thoracic surgery: A prospective study. Medicine (Baltimore) 99(13): e19629.
- Bendixen M, Ole Dan Jørgensen, Christian Kronborg, Claus Andersen, Peter Bjørn Lichtet (2016) Postoperative pain and quality of life after lobectomy via video-assisted thoracoscopic surgery or anterolateral thoracotomy for early stage lung cancer: a randomised controlled trial. Lancet Oncol 17(6): 836-844.
- Bayman EO, Kalpaj R Parekh, John Keech, Atakan Selte, Timothy J Brennan, et al. (2017) A Prospective Study of Chronic Pain after Thoracic Surgery. Anesthesiology 126(5): 938-951.
- Joshi GP, Francis Bonnet, Rajesh Shah, Roseanne C Wilkinson, Frederic Camu, et al, (2008) A systematic review of randomized trials evaluating regional techniques for postthoracotomy analgesia. Anesth Analg 107(3): 1026-1040.
- Seitlinger J, Anne Olland, Sophie Guinard, Gilbert Massard, Pierre-Emmanuel Falcoz (2021) Conversion from video-assisted thoracic surgery (VATS) to thoracotomy during major lung resection: how does it affect perioperative outcomes? Interact Cardiovasc Thorac Surg 32(1): 55-63.
- Fourdrain A, Olivier Georges, Dominique Gossot, Pierre-Emmanuel Falcoz, Jacques Jougon, et al. (2022) Patient risk factors for conversion during video-assisted thoracic surgery-the Epithor conversion score. Eur J Cardiothorac Surg 62(3).
- Heller AR, S Shmygalev, R Sabatowski (2011) Akutschmerzdienst heute: teurer Luxus, notwen- diges Übel oder Methode zur Erlösmaximierung? Anästh Intensivmed 52: 431-441.
- Luketich JD, Stephanie R Land, Erin A Sullivan, Miguel Alvelo-Rivera, Julie Ward, et al. (2005) Thoracic epidural versus intercostal nerve catheter plus patient-controlled analgesia: a randomized study. Ann Thorac Surg 79(6): 1845-1849.
- Campolo M, Dafne Molin, Narinder Rawal, Alfredo Soldati (2012) Protocols to compare infusion distribution of wound catheters. Med Eng Phys 34(3): 326-332.
- Hamilton C, Paul Alfille, Jeremi Mountjoy, Xiaodong Bao (2022) Regional anesthesia and acute perioperative pain management in thoracic surgery: a narrative review. J Thorac Dis 14(6): 2276-2296.
- Shim JG, Kyoung-Ho Ryu, Pyoung On Kim, Eun-Ah Cho, Jin-Hee Ahn, et al. (2020) Evaluation of ultrasound-guided erector spinae plane block for postoperative management of video-assisted thoracoscopic surgery: a prospective, randomized, controlled clinical trial. J Thorac Dis 12(8): 4174-4182.
- Breitling C, M Kretzschmar (2022) [Ultrasound Guided Regional Anesthesia in Thoracic Surgery]. Anasthesiol Intensivmed Notfallmed Schmerzther 57(6): 417-427.
- Steinthorsdottir KJ, Lorna Wildgaard, Henrik Jessen Hansen, René Horsleben Petersen, Kim Wildgaard (2014) Regional analgesia for video-assisted thoracic surgery: a systematic review. Eur J Cardiothorac Surg 45(6): 959-966.
- Hopewell S, Boutron I, Chan AW, Collins GS, de Beyer JA, et al. (2022) An update to SPIRIT and STROBE reporting guidelines to enhance transparency in randomized trials. Nat Med 28(9): 1740-1743.
- Goodyear MD, K. Krleza-Jeric, T Lemmens (2007) The Declaration of Helsinki. BMJ 335(7621): 624-625.
- Mellin-Olsen J, S Staender (2014) The Helsinki Declaration on Patient Safety in Anaesthesiology: the past, present and future. Curr Opin Anaesthesiol 27(6): 630-634.
- Bergmann A, T Schilling (2021) [Intraoperative Ventilation Approaches to One-lung Ventilation]. Anasthesiol Intensivmed Notfallmed Schmerzther 56(5): 329-341.
- Zhang Y, Rong Zhou, Bailing Hou, Suhong Tang, Jing Hao, et al. (2022) Incidence and risk factors for chronic postsurgical pain following video-assisted thoracoscopic surgery: a retrospective study. BMC Surg 22(1): 76.
- Loop T, S Harris, A Grimm (2014) Recent Advances in Postoperative Pain Therapy for Thoracic Surgery. Curr Anesthesiol Reports 4(2): 177-187.
- Manion SC, TJ Brennan (2011) Thoracic epidural analgesia and acute pain management. Anesthesiology 115(1): 181-188.
- Karakaya MA, Davud Yapici, Emre Sertac Bingül, Özlem Turhan, Mert Şentürk (2025) Pro's and con's of different blocks for postoperative analgesia after video-assisted thoracic surgery. Curr Opin Anaesthesiol 38(1): 65-70.
- Flaherty SA (1996) Pain measurement tools for clinical practice and research. AANA J 64(2): 133-140.
- LeBlanc KA, Drake Bellanger, V Keith Rhynes, Mark Hausmann (2005) Evaluation of continuous infusion of 0.5% bupivacaine by elastomeric pump for postoperative pain management after open inguinal hernia repair. J Am Coll Surg 200(2): 198-202.
- Polglase AL, Paul J McMurrick, Paul J B Simpson, Roger J Wale, Peter W G Carne, et al. (2007) Continuous wound infusion of local anesthetic for the control of pain after elective abdominal colorectal surgery. Dis Colon Rectum 50(12): 2158-2167.
- Beaussier M, Hanna El'Ayoubi, Eduardo Schiffer, Maxime Rollin, Yann Parc, et al. (2007) Continuous preperitoneal infusion of ropivacaine provides effective analgesia and accelerates recovery after colorectal surgery: a randomized, double-blind, placebo-controlled study. Anesthesiology 107(3): 461-468.
- Finnerty DT, Aisling McMahon, John R McNamara, Sean D Hartigan, Michael Griffin, et al. (2020) Comparing erector spinae plane block with serratus anterior plane block for minimally invasive thoracic surgery: a randomised clinical trial. Br J Anaesth 125(5): 802-810.
- Tezcan AH, Özgür Karakurt, Mehmet Ali Eryazgan, Semih Başkan, Dilşen Hatice Örnek, et al. (2016) Post-thoracotomy pain relief with subpleural analgesia or thoracic epidural analgesia: randomized clinical trial. Sao Paulo Med J 134(4): 280-284.
- Freise H, HK Van Aken (2011) Risks and benefits of thoracic epidural anaesthesia. Br J Anaesth 107(6): 859-868.






























