Defining Progesterone Profiles and Luteolysis After One or Two Prostaglandin F2α Injections in a 5-Day CIDR-Cosynch Protocol for Suckling Beef Cows
JA Spencer1, K Carnahan2, B Shafii3, WJ Price3, A Ahmadzadeh2*
1Department of Animal Science, Texas A&M AgriLife Extension Services, Texas A&M University, Stephenville, TX 76401, USA
2Department of Animal and Veterinary Sciences, University of Idaho, Moscow, ID, USA
3Statistical Programs, College of Agricultural and Life Sciences, University of Idaho, Moscow, ID, USA
Submission: August 05, 2023; Published: August 22, 2023
*Corresponding author: Amin Ahmadzadeh, Department of Animal and Veterinary Science, 875 Perimeter Dr., MS 2330, Moscow, Idaho, ID, USA. Email: amin@uidaho.edu
How to cite this article: JA Spencer, K Carnahan, B Shafii, WJ Price, A Ahmadzadeh*. Defining Progesterone Profiles and Luteolysis After One or Two Prostaglandin F2α Injections in a 5-Day CIDR-Cosynch Protocol for Suckling Beef Cows. Dairy and Vet Sci J. 2023; 16(1): 555926..DOI: 10.19080/JDVS.2023.16.555926
Abstract
The objective was to examine the effects of one or two doses of conventional prostaglandin F2α (PGF2α), or one high-concentration PGF2α (HighCon) on progesterone (P4) concentrations and luteolysis in suckling beef cows subjected to a 5-day CIDR-Cosynch. On d 0, 54 cows received GnRH (100 μg), and a CIDR was inserted. On d 5, the CIDR was removed and cows were assigned randomly to receive either one dose of PGF2α (1PG, 5 mg/mL, total dose: 25 mg, i.m., n = 19), two injections of conventional PGF2α (2PG, 5 mg/mL, each dose: 25 mg, i.m., n = 17, 12h apart, or one injection of high-concentration PGF2α (HighCon, 12.5 mg/mL, total dose: 25 mg, i.m, n = 18). On d 5, beginning 1 h after CIDR removal, blood samples were collected every 12 h from day 5-8 to measure P4 concentrations. On d 8, cows received a second GnRH injection. Ovaries were examined by ultrasonography on d 0 and 5. A repeated measures generalized linear model was used to determine differences in P4 concentrations. The P4 data were assumed to follow a lognormal distribution. On d 5, all cows had P4 concentrations of >1.0 ng/mL. Serum P4 concentrations decreased over time in all treatments (p < 0.01). There was a treatment by time interaction effect on P4 concentration. Mean P4 concentration tended to differ (p = 0.07) at 60 h between 2PG and 1PG. At 72 h post-treatment, mean P4 tended (p < 0.09) to be lower in 2PG and HighCon compared with 1PG. Regardless of dose, serum P4 concentrations were ≤ 0.5 ng/mL in all treatments. The proportion of cows that went through luteolysis (P4 < 0.5 ng/mL) tended to be greater (p = 0.09) in 2PG and HighCon compared with 1PG (89, 88, and 63%, respectively). The results suggest that one dose of HighCon could be as effective as 2PG in causing luteolysis and decreasing P4 by the time of AI in suckling beef cows. Considering P4 concentrations and its relationship to fertility, compared with two injections, the administration of one high concentration PGF2α is more practical for synchronizing beef cows, as it reduces animal handling, labor, and drug costs, while it improves compliance.
Keywords: Synchronization; Prostaglandin dose; Progesterone profile; Cattle
Abbreviations: CIDR: Controlled Internal Drug Release; CL: Corpus Luteum; BCS: Body Condition Score; GLM: Generalized Linear Model
Introduction
Estrus synchronization protocols that result in greater synchronized ovulation and pregnancy per AI (P/AI) will facilitate the use of AI and may increase its adoption among beef producers. The use of Cosynch protocols have allowed for synchronization of ovulation and timed AI (TAI), as well as reduced number of handlings when compared to Ovsynch [1]. Addition of progesterone (P4) in the form of controlled internal drug release (CIDR) inserts between the initial GnRH and prostaglandin F2α (PGF2α) injections (7-d CIDR-Cosynch) improve synchronization, and P/AI by preventing premature estrus and ovulation before PGF2α [2-4].
Reducing the duration of CIDR treatment to the induction of luteolysis from 7 to 5-day in the CIDR-Cosynch, has been shown to increase P/AI in beef cows [5]. The authors, however concluded that, two injections of PGF2α (2PG, 7 to 24 h apart) on d 5 were required to reliably induce luteolysis by the time of AI in 5-day CIDR-Cosynch [5]. Other studies have also shown an improvement in P/AI in beef cows receiving 2PG compared with one injection of PGF2⍺ (1PG) in a 5-day CIDR-Cosynch protocol [6,7]. It was hypothesized that improved P/AI may be attributable to efficacy of 2PG in causing complete luteolysis and lowering P4 concentrations by the time of AI compared with 1PG. Interestingly, P4 concentrations following PGF2α, and ovarian status were not assessed in any of these published studies to allow comparison of 1PG vs. 2PG effects on P4 and ovarian status at the time of AI. Thus, it is not clear whether the observed improvement in P/AI in these studies is attributable to effectiveness of 2PG in causing luteolysis or is due to other factors. The addition of a second PGF2⍺ treatment in a 5-day CIDR-Cosynch protocol for beef cows adds extra cow handling, increases cost, labor, and time requirements, and potentially decreases protocol compliance. In a 5-dayCIDRCosynch protocol, the results of our study [8] showed that one 25 mg dose of high concentration PGF2α (HighCon, dinoprost tromethamine, 12.5 mg/mL, Zoetis Inc.) administered at CIDR removal is as effective as 2PG (5 mg/mL) at inducing luteolysis and thus reducing P4 to optimal concentrations (P4 < 0.5 ng/mL) by the time of AI in dairy cows. According to the manufacturer, HighCon reaches a greater maximum plasma concentration (Cmax), and perhaps undergoes less blood level fluctuations following administration than the standard 25 mg dose of dinoprost tromethamine PGF2⍺ [9].
There is a lack of sufficient evidence on the effects of 1PG vs. 2PG on luteal function and blood P4 in a 5-day CIDR-Cosynch protocol. Furthermore, the HighCon effect on P4 profile is unknown in beef cattle. Based on our previous finding in dairy cows [8], it is plausible that one does of HighCon is as effective in causing luteolysis as 2PG hence potentially reduce above mentioned difficulties associated with 2PG injections. The objectives of this study were to examine the effects of 1PG, 2PG (12 h apart), and one high concentration, HighCon PGF2α on luteolysis and P4 profiles in suckling beef cows subjected to a 5-day CIDR-Cosynch protocol.
Materials and Methods
All procedures and protocols followed the University of Idaho, Animal Care and Use Committee. Thirty-one suckling Charolais beef cows from the University of Idaho (Moscow, Idaho) research facility were used for this study. All cows were on average 58 days postpartum at the initiation of the study.
Initially, 78 suckling Charolais beef cows were enrolled into the study and synchronized for the first insemination using a 5-day CIDR-Cosynch protocol. Twenty-four cows were removed as they did not have a CL and (or) elevated P4 concentrations before treatment (3 cows), or they were acyclic as blood P4 from day -7, 0, and 5 were all < 0.5 ng/mL (21 cows). Cows were housed on pasture and had access to supplemental hay, a mineral supplementation, and ad libitum access to water.
Experimental design and treatments

Seven days before estrus synchronization, jugular blood samples were collected from all cows (Figure 1). On d 0 (initiation of synchronization), all cows received GnRH (100 μg, i.m., Factrel, Fort Dodge Animal Health, Fort Dodge, IA), and a blood sample was obtained. Simultaneously, a CIDR (1.38 g P4, Eazi-Breed CIDR, Zoetis, Florham Park, NJ) was inserted (Figure 1), and all cows were subjected to transrectal ultrasonography (Aloka SSD-500 V, Aloka, Tokyo, Japan), and categorized into two groups based on presence or absence of a corpus luteum (CL). On the same day body condition score (BCS) was assessed using the average of two evaluators (scale 1-9 with 1 = emaciated and 9 = obese) [10]. Five days later CIDR inserts were removed, and cows were stratified by days postpartum, parity, and presence or absence of a CL on day 0, and presence of one or two CL on day 5 (existing CL and new CL; follicle ovulated to initial GnRH) and assigned randomly to one of three treatments: one PGF2α injection (1PG, n = 19, 25 mg, i.m., Lutalyse, Zoetis, Florham Park, NJ), two PGF2α injections (2PG, n = 17, 50 mg total, i.m., Lutalyse, Zoetis, Florham Park, NJ) given 12 h apart, or one high concentration PGF2α (HighCon, n = 18, 25 mg, i.m., Lutalyse HighCon, Zoetis, Florham Park, NJ) (Figure 1). Before treatment and 60 min after CIDR removal, blood samples were taken, and ultrasonography was conducted to confirm the presence of luteal tissue. Following treatment, blood samples were collected every 12 h from day 5 to 8 to measure P4 concentrations (Figure 1). Between day 5 and 8, cows were monitored every 4 to 6 h for estural behavior as well as removal of tail chalk painting. Regardless of expression of estrus, all cows received a second GnRH (100 μg) on day 8 (Figure 1). Two cows from the 2PG group were eliminated from study because of complications unrelated to the study.
Ovarian examination
On day 0 and 5 transrectal ultrasonography (Aloka SSD-500 V, Aloka, Tokyo, Japan) was conducted to assess ovarian structures. Cows were categorized as developing a new CL or possessing an old CL at the time of treatment. The presence of a dominant follicle (≥ 10 mm diameter) on d 0, and the presence of a CL on d 5 in the same location identified cows (n = 31) that ovulated to the initial GnRH and formed a new CL. Cows presenting a CL on day 0 and day 5 in the same location were classified as possessing an old CL at the time of treatment. Luteolysis was defined as P4 concentrations ≥ 0.5 ng/mL on day 5, and P4 < 0.5 ng/mL on day 8.
Blood samples and progesterone quantification
Blood samples were collected via jugular venipuncture using an 18-gauge, 1½” single use blood collection needle. Blood samples were collected using a 10 mL vacuumed tube (Covidien LLC, Mansfield, MA). All samples were placed on ice and stored at 4°C for 18 to 24 h. Samples were then centrifuged for 20 min at 2,400 × g and 4°C. Serum was harvested and stored at -20°C until assayed for P4 concentrations. Progesterone concentrations were analyzed using a double antibody, RIA (MP Biomedicals, Costa Mesa, CA) under equilibrium conditions. The standard curve ranged from 0.05 to 25 ng/mL, and all samples and standards were run in duplicates, with an intra- and inter-assay coefficient of variance of 3.6% and 6.7%, respectively.
Statistical analysis
The differences in days postpartum and BCS between treatment groups were assessed using a generalized linear model (GLM) procedure in SAS (v. 9.4, SAS® Institute Inc.) [11]. The analysis of variance was also used to determine differences on day 0, 5, and 8 P4 concentrations. The model included the main effect of treatment. A univariate analysis was conducted indicating a nonnormal distribution for P4 data. A repeated measure generalized linear mixed model [11] was used to determine differences in P4 concentrations across time assuming a lognormal distribution [12]. The model included the fixed effects of treatment, time, and the time by treatment interaction. Initial P4 concentration prior to treatment (day 5) were also included in the analysis as a covariate. Cows within treatments were considered as random effects, and the correlation structure for the repeated measures was ARMA (1,1).
The differences in proportion of cows between treatments with the presence of a CL on d 0 and proportion of cows with luteolysis on d 8 were analyzed using a generalized linear model (GLIMMIX), assuming a binomial distribution. Non-orthogonal predetermined contrasts were made between groups for ovarian structures on day 0 (presence or absence of CL), day 5 (development of new CL), and incidence of luteolysis (60 and 72 h after treatments). Using Bonferroni adjusted for multiple comparison [13], differences between 1PG vs. 2PG, 1PG vs. HighCon, and 2PG vs. HighCon were examined. All statistical computations were carried out using SAS (v. 9.4, SAS Institute Inc. 2015). Significance was declared at p < 0.05 and a tendency at p ≤ 0.1.
Results and Discussion
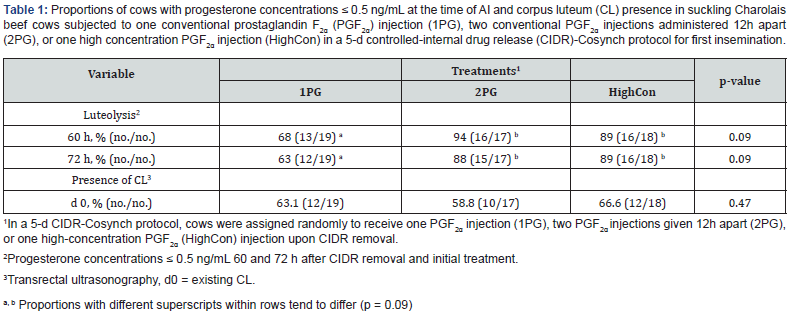
Mean days postpartum and BCS were 69 ± 3 and 6.3 ± 0.1, respectively. There were no differences detected in BW (p = 0.48) between treatments and averaged 682.7 ± 28.6 kg. The proportion of cows with an existing CL on day 0 of the experiment did not differ (p = 0.47) between treatments (Table 1). By experimental design, all cows had CL on day 5 and the proportion of cows that ovulated to the initial GnRH did not differ (p = 0.35) between treatments. It should be noted that the probability of detecting a small difference in incidence of ovulation between treatments in this experiment was limited by the number of cows enrolled. By design and based on P4 concentrations on day 0 or 5, all cows were cyclic and had a CL before administration of treatment on day 5.
Mean P4 concentrations on day 0 (initiation of synchronization) did not differ between treatments (p = 0.37) and were 0.90 ± 0.38 for 1PG, 1.20 ± 0.40 for 2PG, and 1.64 ± 0.39 ng/mL for HighCon, respectively (Figure 2). There was no difference in mean P4 concentrations between treatments on day 5 (p = 0.50) and were 2.39 ± 0.43, 1.74 ± 0.45, and 1.77 ± 0.44 ng/mL for 1PG, 2PG, and HighCon, respectively (Figure 2).

The P4 concentrations from 12 to 72 h after treatments were skewed indicating a non-normal distribution. Hence, P4 data were assumed to follow a lognormal distribution. Progesterone concentrations on day 5 were used as a covariate in the model to account for individual P4 concentration variation before treatment. As expected, P4 concentrations decreased over time (p < 0.01) in all treatments. Mean P4 concentrations decreased to < 1.0 ng/mL across treatment within the first 12 h after treatment and remained below 1.0 ng/mL throughout the experiment, however, there was an effect of treatment by time on serum P4 concentrations (p = 0.02) as P4 profiles differ over time between treatments (Figure 3). Mean serum P4 concentrations tended to differ (p = 0.07) at 36 and 60 h between 2PG and 1PG. At 72 h after treatment (around the time of AI in a 5-day CIDR-Cosynch protocol), mean P4 concentrations tended (p = 0.06) to be lesser in 2PG and HighCon compared with 1PG but 2PG and HighCon did not differ. Mean serum P4 concentrations were 0.50 ± 0.11, 0.18 ± 0.12, and 0.25 ± 0.11 ng/mL for 1PG, 2PG, and HighCon, respectively (Figure 2 and 3). Mean P4 concentrations, however, were ≤ 0.5 ng/mL in all treatments by 72 h, the time of AI in a 5-day CIDR-Cosynch protocol.
The differences in P4 concentrations at 60 and 72 h (day 8) after treatments were further examined to determine the incidence of luteolysis (Table 1). The proportion of cows with luteolysis (P4 < 0.5) at both 60 and 72 h for HighCon and 2PG tended to be greater than 1PG (p = 0.09). Interestingly, the incidence of luteolysis showed no difference between 2PG and HighCon (Table 1).
There are several factors during a TAI estrus synchronization protocol that greatly influence pregnancy outcome. One important factor is the ability of PGF2α to cause complete luteolysis within 48 h after administration, hence optimal P4 concentrations by the time of AI [14]. The inability of PGF2α to successfully induce luteolysis and reduce P4 to ≤ 0.5 ng/mL by the time of AI has been termed incomplete luteolysis [15,16]. The improved P/AI in the 5-day CIDR-Cosynch depends on complete luteolysis by the time of AI using 2PG [5-7]. The authors hypothesized that the GnRHinduced newly formed CL, is refractory to one PGF2α, hence two injections of PGF2α (7 to 24 h apart) on day 5 were required to reliably induce luteolysis by the time of AI in 5-day CIDR-Cosynch, but in those studies the effect 1PG vs. 2PG on luteolysis and or P4 profile were not studied. As indicated, the addition of a second PGF2⍺ adds additional cattle handling, increases cost, labor, and time requirements, and potentially decreases protocol compliance. Can one injection of HighCon be as effective as 2PG in reducing P4 concentrations and induce luteolysis because of its different pharmacokinetics? The results of current study provide evidence, for the first time, that administration one dose of HighCon is as effective as two doses of conventional PGF2α, in reducing P4 concentrations and causing luteolysis by the time AI in 5-day CIDR-Cosynch. It should be noted that although one PGF2α does not appear to be as effective as two PGF2α or one HighCon PGF2α in rapidly reducing P4 concentrations, the P4 concentrations in all treatments were ≤ 0.5 ng/mL on d 8 when AI usually occurs. These results are like our previous findings [8] that showed one injection of HighCon was as effective in reducing P4 as 2PG injection in dairy cows. Also, Ahmadzadeh et al. [17] investigated the effects of one PGF2α (25 mg), 1.5 PGF2α (37.5 mg) administered once, or two 0.5 PGF2α (25 mg total) injections given 7 h apart following CIDR removal in a 5-day CIDR-Cosynch protocol in beef cows. These authors found no difference in P4 concentrations at the time of AI regardless of treatment, and all treatments had mean P4 ≤ 0.5 ng/ mL.

Currently, there is a limited amount of evidence about the effectiveness of one or two PGF2α injections on P4 concentrations for suckling beef cows subjected to a 5-day CIDR-Cosynch protocol. The ability of PGF2α to cause complete luteolysis and reduce P4 concentrations to less than 0.5 ng/mL by the time of AI does aid in maximizing the fertility of beef cows replace with [18]. Any timed-AI program, including the 5-day CIDR, has limitations because of various factors, one of which being the induction of luteolysis in all luteal structures in response to PGF2α [16]. It is suggested that the concentrations of P4 after PGF2α administration should approach to < 0.4 ng/mL before time-AI to increase the odds of GnRH-induced ovulation as well as and subsequent pregnancy outcome. Our investigation shows that one dose of HighCon is just as effective as two doses of traditional PGF2α in lowering P4 while lowering the labor and compliance costs related to handling animals. To our knowledge only one study examined P4 concentrations at PGF2α and AI to determine luteolysis in beef cows using 5-day CIDR-Cosynch [19]. In that study, beef cows were treated with one PGF2α, a double dose (50 mg) of PGF2α, or two PGF2α injections administered 8 h apart in a 5-day CIDR-Cosynch protocol. These researchers found that luteolysis tended to differ between groups with two PGF2α injections (97%) 8 h apart having greater luteolysis when compared to a double dose (88%) or one PGF2α (87%) injection at CIDR removal. In that study, luteolysis was defined as P4 concentrations > 2 ng/mL at CIDR removal and < 1 ng/mL at the time of AI.
Although the total dose of dinoprost tromethamine were the same (25 mg), the effectiveness of one dose of HighCon compared to one injection of conventional PGF2α on luteolysis cannot be elucidated from this study. This difference is less likely related to the difference in P4 concentrations or luteal structure of the cows before treatment as incidence of luteolysis and serum P4 concentrations were not different between HighCon and 1PG. The difference in the blood pharmacokinetics between HighCon and conventional PGF2α, as described by Zoetis (2015) may be the contributing factor.
Conclusion
Considering P4 concentrations and its relationship to fertility, the administration of one PGF2α is more practical for synchronizing beef cows, as it reduces animal handling, labor, and drug costs. Further, the addition of a second PGF2α treatment within the 5-day CIDR-Cosynch program increases labor associated with animal handlings, and may decrease protocol compliance, ultimately reducing the potential benefits of two PGF2α injections in a 5-day CIDR-Cosynch protocol. Future research is needed to compare the effects of one injection of HighCon with two PGF2α on P/AI to further support the use of one high concentrated PGF2α injection in a 5-day CIDR-Cosynch protocol for suckling beef cows.
Acknowledgement
The authors wish to express their appreciation to the staff at the University of Idaho, Beef Research Center, and thank Zoetis (Florham Park, NJ) for donating Lutalyse® and CIDR®. The research was also supported, in part, by the Idaho Agriculture Experimental Station Hatch Formula Funding.
References
- Geary TW, Pas JW, Thrift FA, Dolezal SL (1998) Effects of timed insemination following synchronization of ovulation using the Ovsynch or CO-synch protocol in beef cows. Prof Anim Sci 14(4): 217-220.
- Larson JE, Lamb GC, Stevenson JS, Johnson SK, Day ML, et al. (2006) Synchronization of estrus in suckled beef cows for detected estrus and artificial insemination and artificial insemination using gonadotropin-releasing hormone, prostaglandin F, and progesterone. J Anim Sci 84(2): 332-342.
- Lamb GC, Stevenson JS, Kesler DJ, Garverick HA, Brown DR, et al. (2001) Inclusion of an intravaginal progesterone insert plus GnRH and prostaglandin F2α for ovulation control in postpartum suckled beef cows. J Anim Sci 79(9): 2253-2259.
- Gunn PJ, Culp KC, Lemenager RP, Bridges GA (2016) Efficacy of the 5-day CO-Synch ovulation synchronization protocol with or without the inclusion of exogenous progesterone in beef cows. Prof Anim Sci 3: 82-89.
- Bridges GA, Helser LA, Grum DE, Mussard ML, Gasser CL, et al. (2008) Decreasing the interval between GnRH and PGF2α from 7 to 5 days and lengthening proestrus increases timed-AI pregnancy rates in beef cows. Theriogenology 69: 843-851.
- Kasimanickam R, Day ML, Rudolph JS, Hall JB, Whittier WD (2009) Two doses of prostaglandin improve pregnancy rates to timed-AI in a 5-day progesterone-based synchronization protocol in beef cows. Theriogenology 71(5): 762-767.
- Whittier WD, Kasimanickam RK, Currin JF, Schramm HH, Vlcek M (2010) Effect of timing of second prostaglandin F 2 alpha administration in a 5-day, progesterone-based CO-Synch protocol on AI pregnancy rates in beef cows. Theriogenology 74(6): 1002-1009.
- Spencer JA, Carnahan K, Price W, Shafii B, Ahmadzadeh A (2018) One injection of high-concentration prostaglandin F2α is as effective as two injections of conventional prostaglandin F2α in causing luteolysis for dairy cows subjected to a 5-d CIDR-Cosynch protocol. J Dairy Sci 101: 288.
- Zoetis Animal Health (2015) Lutalyse (dinoprost tromethamine injection) product insert. Florham Park (NJ): Zoetis Inc.
- Diskin MG, Kenny DA (2016) Managing the reproductive performance of beef cows. Theriogenology 86: 379-387.
- SAS Institute. SAS 9.4, Version 2. 2015, SAS Institute Inc. Cary, NC.
- Stroup WW (2015) Rethinking the Analysis of Non-Normal Data. Plant and Soil Science. Agron J 106: 1-17.
- Games PA (1971) Multiple comparisons of means. American Ed Research J 8: 531-565.
- Stevenson JS (2016) Physiologcal predictors of ovulation and pregnancy risk in a fixed-time artificial insemination program. J Dairy Sci 99: 10077-10092.
- Brusveen DJ, Cunha AP, Silva CD, Cunha, PM, Sterry RA, et al. (2008) Altering the time of the second gonadotropin-releasing hormone injection and artificial insemination (AI) during Ovsynch affects pregnancies per AI in lactating dairy cows. J Dairy Sci 91: 1044-1052.
- Carvalho PD, Souza AH, Wiltbank MC, Fricke PM (2015) Progesterone concentration at each treatment during an Ovsynch protocol affects fertility to timed AI in lactating Holstein cows. J Dairy Sci 98(Suppl. 2): 92.
- Ahmadzadeh, A, Carnahan K, Robison T, Autran C (2011) Effect of various doses of prostaglandin F2α on behavior and blood progesterone in beef cows. J Anim Sci 89(E-Suppl 1): 253.
- Stevenson J S, Lamb GC (2016) Contrasting effects of progesterone on fertility of dairy and beef cows. J Dairy Sci 99: 5951-5964.
- Bridges GA, Ahola JK, Brauner C, Cruppe LH, Currin JC, et al. (2012) Determination of the appropriate delivery of prostaglandin F2α in the five-day CO-Synch+ controlled intravaginal drug release protocol in suckled beef cows. J Anim Sci 90: 4814-4822.






























