Abstract
Objective: This study aims to assess the therapeutic effects and safety profile of dydrogesterone in managing dysmenorrhea secondary to endometriosis.
Methods: A retrospective analysis was conducted on 30 patients with endometriosis-induced dysmenorrhea treated at the Department of
Gynecology, Second People’s Hospital of Changzhou, Nanjing Medical University, between January 2022 and September 2024. All patients were
diagnosed with endometriosis via ultrasound or surgical pathology and exhibited dysmenorrhea symptoms. They received dydrogesterone (10
mg orally, twice daily from day 5 to day 25 of the menstrual cycle) for six consecutive menstrual cycles. Pain was evaluated using the Visual
Analog Scale (VAS) before and after treatment, and menstrual characteristics and adverse reactions were documented.
Results: Dysmenorrhea symptoms improved significantly, with a marked reduction in VAS scores (P < 0.01). Menstrual duration was shortened,
and menstrual bleeding volume decreased. No patients required additional analgesics, and only three experienced mild gastrointestinal
reactions.
Conclusion: Dydrogesterone is a safe and effective treatment for dysmenorrhea caused by endometriosis, with minor adverse effects.
Keywords: Dydrogesterone; Endometriosis; Dysmenorrhea; Visual Analog Scale (VAS)
Introduction
Endometriosis is a prevalent gynecological condition characterized by the presence of endometrial tissue outside the uterine cavity, affecting approximately 10% of women of reproductive age[1]. This condition is associated with a range of symptoms, including dysmenorrhea, chronic pelvic pain, and infertility, significantly impacting the quality of life [2]. Dysmenorrhea, in particular, is a hallmark symptom of endometriosis, experienced by a majority of affected women [3]. The pathophysiology of endometriosis-related pain is complex, involving inflammation, abnormal nerve growth, and altered pain processing mechanisms. The experience of pain in endometriosis is complex and multifactorial, arising not only from classic nociceptive mechanisms (e.g., inflammation, tissue damage) but also involving neuropathic and oncoplastic components, the latter being characterized by altered central nervous system pain processing [4]. Traditional treatments for endometriosis-related pain include nonsteroidal antiinflammatory drugs (NSAIDs), oral contraceptives, and traditional Chinese medicine [5]. While these treatments offer some relief, they often come with significant side effects or limited efficacy. For instance, NSAIDs can cause gastrointestinal discomfort, and oral contraceptives may not be suitable for all women, especially those planning to conceive. Therefore, there is a need for alternative treatments that are both effective and safe [6].
Dydrogesterone, a selective progesterone receptor modulator (SPRM), has emerged as a promising therapeutic option for managing endometriosis-related pain [7]. It works by binding to the progesterone receptor with high specificity, inhibiting endometrial proliferation, and reducing prostaglandin production, thereby alleviating pain symptoms [8]. Clinical trials have shown that dydrogesterone can significantly reduce pain intensity and improve the quality of life in women with endometriosis. For example, a recent study found that dydrogesterone treatment led to a marked reduction in VAS scores, a widely used measure of pain intensity [9]. Additionally, dydrogesterone has been shown to be effective in reducing endometriosis-associated pain without causing significant bone loss, which is a concern with some other treatments like gonadotropin-releasing hormone agonists (GnRH-a) [10]. While previous randomized trials have demonstrated the efficacy of dydrogesterone, there remains a need for real-world evidence regarding its use in routine clinical practice. This retrospective cohort study aims to address this gap by specifically investigating the clinical efficacy and safety of dydrogesterone in treating endometriosis-associated dysmenorrhea. We focused on evaluating changes in pain intensity (measured by VAS scores) and menstrual duration, alongside monitoring its safety profile.
Materials and Methods
General Data
This study included 30 patients aged 18-50 years with endometriosis-induced dysmenorrhea, diagnosed by ultrasound or surgical pathology. Patients had not used hormonal treatments in the preceding three months and were capable of assessing pain intensity. Among the 30 patients, the types of endometrioses were classified as follows: ovarian endometriosis (n=21), deep infiltrating endometriosis(n=0), and peritoneal endometriosis (n=7). For patients with endometriosis, the involved sites included the rectum (n=1), vagina (n=1), and uterosacral ligaments (n=0). Exclusion criteria included inability to follow up, contraindications to the study drug, and comorbidities such as cardiovascular or gastrointestinal diseases. As this was a retrospective study, a formal sample size calculation was not conducted. The sample included all consecutive eligible patients who presented to our department during the study period and met the inclusion criteria. This retrospective study was approved by the Clinical Medical Technology Ethics Committee of the Second People’s Hospital of Changzhou (Nanjing Medical University; approval No. YLJSA201, dated 12 January 2022). In accordance with the Declaration of Helsinki and local regulations, the requirement for written informed consent was waived because the investigation involved analysis of anonymized data retrieved from routine clinical records.
Methods Treatment Protocol
All patients with dysmenorrhea received oral dydrogesterone tablets (Dafutong, Abbott Laboratories, USA, 10 mg tablets) for treatment. The regimen involved taking 10 mg of dydrogesterone orally twice daily from days 5 to 25 of the menstrual cycle, with the course lasting six menstrual cycles. No oral steroid analgesics were administered during the treatment period.
Observation Indicators
Pain was assessed using the VAS, with scores ranging from 0 (no pain) to 10 (worst pain). The VAS is a widely used and validated tool for measuring pain intensity in clinical studies. Menstrual duration and bleeding volume were recorded at each follow-up visit. Adverse reactions were monitored throughout the study period to assess the safety profile of dydrogesterone. The primary outcomes measured were changes in VAS scores and menstrual duration, while secondary outcomes included the incidence and severity of adverse reactions.
Statistical Analysis
Data were analyzed using SPSS 12.0 software (IBM Corp., Armonk, NY, USA). The primary outcome measures were changes in pain intensity, as assessed by the Visual Analog Scale (VAS) scores, and menstrual duration. To evaluate the efficacy of dydrogesterone treatment, paired t-tests were conducted to compare the mean VAS scores and menstrual duration before and after treatment at each time point (1 month, 3 months, and 6 months). The assumption of normality was verified using the Shapiro-Wilk test, and the homogeneity of variances was confirmed using Levene’s test. All tests were two-tailed, and a P-value of less than 0.05 was considered statistically significant. Additionally, effect sizes were calculated using Cohen’s d to quantify the magnitude of the treatment effects. Since all patients completed the study, no missing data were present.
Results
All 30 patients completed the study. The baseline characteristics of the patients are summarized in (Table 1). The mean age of the patients was 32 years (range 18-50 years), with an average menstrual duration of 5.0 days (range 5-8 days). The average number of pregnancies was 3 (range 0-5), and the average number of deliveries was 1 (range 0-2). Pain Assessment Using Visual Analog Scale (VAS) (Table 2) ,Pretreatment, the severity of dysmenorrhea was categorized as follows: 9 patients had mild dysmenorrhea (VAS score 1-3), 17 had moderate dysmenorrhea (VAS score 4-6), and 4 had severe dysmenorrhea (VAS score 7-10). The mean VAS score before treatment was 85 ± 15.4, indicating a high level of pain intensity among the study population. After initiating dydrogesterone treatment, there was a significant reduction in VAS scores across all time points. Specifically, the mean VAS score decreased to 66 ± 12.6 after 1 month (P < 0.05), to 45 ± 9.9 after 3 months (P < 0.05), and to 39 ± 12.6 after 6 months (P < 0.05). This progressive reduction in VAS scores indicates a substantial alleviation of pain symptoms over the treatment period. In addition to pain relief, the treatment also resulted in a significant reduction in menstrual duration (Table 3). The mean menstrual duration before treatment was 6.0 ± 1.4 days. After treatment, the mean menstrual duration decreased to 5.0 ± 1.8 days after 1 month (P < 0.05), to 4.5 ± 0.9 days after 3 months (P < 0.05), and to 3.0 ± 1.6 days after 6 months (P < 0.05). Regarding safety and tolerability, no cases of amenorrhea were reported by any of the 30 patients at the 6-month follow-up. This aligns with the progestogenic mechanism of dydrogesterone, which generally preserves the menstrual cycle. Therefore, we have not described it in the table.
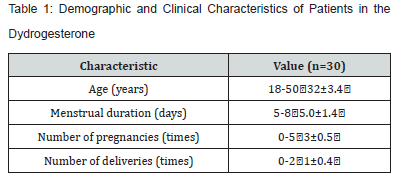
Values are expressed as number, mean ± standard deviation or percentage.
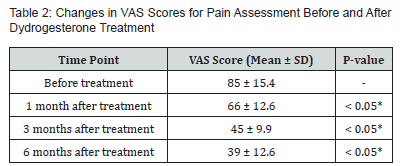
Values are expressed as number, mean ± standard deviation or percentage. *P<0.05
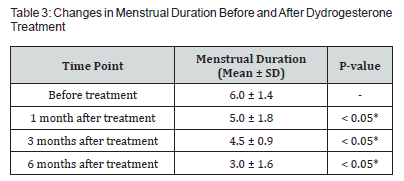
Values are expressed as number, mean ± standard deviation or percentage. *P<0.05
Discussion
Endometriosis is a multifactorial gynecological disorder characterized by the presence of endometrial-like tissue outside the uterus, leading to chronic pelvic pain, dysmenorrhea, and infertility [11]. The pathogenesis of endometriosis is complex and not fully understood, involving retrograde menstruation, coelomic metaplasia, genetic predisposition, and immune dysfunction [12]. Traditional treatments for endometriosisrelated pain include nonsteroidal anti-inflammatory drugs (NSAIDs), combined oral contraceptives, progestins, and gonadotropin-releasing hormone agonists (GnRH-a). However, these treatments may have significant side effects or limited efficacy [13]. Dydrogesterone, a selective progesterone receptor modulator (SPRM), has shown promising therapeutic effects in managing endometriosis-related pain [14]. It works by binding to the progesterone receptor with high specificity, inhibiting endometrial proliferation, and reducing prostaglandin production, thereby alleviating pain symptoms. In a multicenter clinical study, dydrogesterone significantly reduced the total dysmenorrhea score and related symptoms in Japanese women with dysmenorrhea, with a statistically significant decrease observed after the second menstrual cycle [15]. Similar findings were observed in our study, where dydrogesterone treatment led to a marked reduction in VAS scores and menstrual duration, indicating its efficacy in relieving dysmenorrhea caused by endometriosis.
Moreover, dydrogesterone has several advantages over other treatments. Unlike some synthetic progestins, dydrogesterone does not have significant androgenic or estrogenic activity, resulting in fewer side effects [16]. It also maintains ovulation and fertility, making it a suitable option for women who wish to conceive. In addition, dydrogesterone has been shown to be effective in reducing endometriosis-associated pain without causing significant bone loss, which is a concern with some other treatments like GnRH-a. Clinical studies have shown that medical treatment can indeed alleviate dysmenorrhea symptoms. However, recurrence of pain symptoms after discontinuation of treatment is also common, with reports indicating that 17% to 34% of women who have received treatment experience this. Nevertheless, continuing treatment to achieve sustained symptom relief may be limited by drug intolerance or an increased risk of adverse events. There is a need for prospective data on the long-term efficacy and safety of different types of medical treatments for endometriosis to determine the optimal and longest duration of treatment. Recent insights into endometriosis have underscored the necessity for more personalized and multimodal treatment strategies [17]. Viewing endometriosis as a multi-system disorder highlights the need to address its diverse symptoms and comorbidities. For instance, the increased biosynthesis of inflammatory mediators is now recognized as a pivotal driver of disease progression and pain, prompting the development of targeted therapies. Additionally, emerging research on the gut–microbiota–brain axis suggests new potential avenues for managing endometriosis-associated symptoms [18]. These advancements reflect a shift towards more holistic and targeted approaches in treating endometriosis.
Despite the promising results, our study has several limitations. Firstly, the sample size was relatively small, with only 30 patients included. This limits the statistical power and generalizability of our findings. Secondly, our study did not include a comparison with other treatment modalities, such as NSAIDs, oral contraceptives, or GnRH agonists. This makes it difficult to definitively conclude that dydrogesterone is superior or non-inferior to other available treatments [19- 21]. We fully acknowledge these limitations inherent in this initial retrospective analysis. We have initiated a prospective randomized controlled trial (RCT) that aims to enroll a larger patient cohort and includes parallel control groups receiving NSAIDs, oral contraceptives, and GnRH agonists. We anticipate that this ongoing research will provide more robust evidence regarding the comparative efficacy of dydrogesterone.
Conclusion
In summary, dydrogesterone is a safe and effective treatment option for dysmenorrhea caused by endometriosis, with minimal adverse effects. Its mechanism of action and favorable side effect profile make it a valuable addition to the therapeutic arsenal for managing this challenging condition. Future research should focus on large-scale, multicenter, randomized controlled trials to further validate the efficacy and safety of dydrogesterone in different patient populations. Additionally, exploring the potential of novel therapeutic strategies, such as targeted anti-inflammatory therapies and microbiome interventions, could lead to more personalized and effective treatments for endometriosis.
Acknowledgments
We would like to express our gratitude to all those who helped us during the writing of this manuscript.
Availability of Data and Materials. The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Funding
This project was supported by the research project :2023 Changzhou Municipal Health Commission Major Science and Technology Project: Mechanism of Estrogen-Mediated LINC01541 Regulation of miR-429 in Promoting Angiogenesis in Endometriosis (Grant No.: ZD202343).
Research Ethics and Informed Consent
All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of the Second People’s Hospital of Changzhou, affiliated with Nanjing Medical University (approval number: 202123).
References
- Buck Louis GM, Hediger ML, Matthew Peterson C, Croughan M, Sundaram R, et al., (2011) Incidence of endometriosis by study population and diagnostic method: the ENDO study. Fertil Steril 96(2): 360-365.
- Eskenazi B and ML Warner, (1997) Epidemiology of endometriosis. Obstet Gynecol Clin North Am 24(2): 235-258.
- Maulenkul T, Kuandyk A, Makhadiyeva D, Dautova A, Terzic M, et al., (2024) Understanding the impact of endometriosis on women's life: an integrative review of systematic reviews. BMC Womens Health 24(1): 524.
- Raimondo D, Raffone A, Renzulli F, Sanna G, Raspollini A, et al., (2023) Prevalence and Risk Factors of Central Sensitization in Women with Endometriosis. J Minim Invasive Gynecol, 30(1): 73-80.e1.
- Della Corte L, Di Filippo C, Gabrielli O, Reppuccia S, Lucia La Rosa V, et al., (2020) The Burden of Endometriosis on Women's Lifespan: A Narrative Overview on Quality of Life and Psychosocial Wellbeing. Int J Environ Res Public Health 17(13): 4683.
- Schliep KC, Mumford SL, Peterson CM, Chen Z, Johnstone EB, et al., (2015) Pain typology and incident endometriosis. Hum Reprod 30(10): 2427-2438.
- Tsai PJ, Lin YH, Chen JL, Yang SH, Chen YC, et al., (2017) Identifying Chinese Herbal Medicine Network for Endometriosis: Implications from a Population-Based Database in Taiwan. Evid Based Complement Alternat Med 2017: 7501015.
- Mazur-Bialy A, Tim S, Pępek A, Skotniczna K, Naprawa G, et al., (2024) Holistic Approaches in Endometriosis - as an Effective Method of Supporting Traditional Treatment: A Systematic Search and Narrative Review. Reprod Sci 31(11): 3257-3274.
- Peng C, Y Huang, Y Zhou, (2021) Dydrogesterone in the treatment of endometriosis: evidence mapping and meta-analysis. Arch Gynecol Obstet 304(1): 231-252.
- Carvalho BR (2022) Dydrogesterone as an Option in the Medical Treatment of Endometriosis: A Brief Comment. Rev Bras Ginecol Obstet 44(8): 802-803.
- Shahab M, Ziyu P, Waqas M, Zheng G, Bin Jardan YA, et al., (2024) Targeting human progesterone receptor (PR) through pharmacophore-based screening and molecular simulation revealed potent inhibitors against breast cancer. Sci Rep 14(1): 6768.
- Xue HL, WJ Hao, B Wang, (2023) A comparative study between Dydrogesterone alone and combined with Non-Steroidal Anti-Inflammatory Drugs in the treatment of Mild Endometriosis. Pak J Med Sci 39(5): 1406-1410.
- Tang W, Zhu X, Bian L, Zhang B, et al., (2024) Research progress of dydrogesterone in the treatment of endometriosis. Eur J Obstet Gynecol Reprod Biol 296: 120-125.
- Chapron C, et al., (2019) Rethinking mechanisms, diagnosis and management of endometriosis. Nat Rev Endocrinol 15(11): 666-682.
- Chen Y, S Waseem, L Luo, (2025) Advances in the diagnosis and management of endometriosis: A comprehensive review. Pathol Res Pract 266: 155813.
- Morris SA, KS Korach, KA Burns, (2021) Unique Sensitivity of Uterine Tissue and the Immune System for Endometriotic Lesion Front Physiol 12: 805784.
- Becker CM, Gattrell WT, Gude K, Singh SS (2017) Reevaluating response and failure of medical treatment of endometriosis: a systematic review. Fertil Steril 108(1): 125-136.
- Sukhikh GT, Adamyan LV, Dubrovina SO, Baranov II, Bezhenar VF, et al., (2021) Prolonged cyclical and continuous regimens of dydrogesterone are effective for reducing chronic pelvic pain in women with endometriosis: results of the ORCHIDEA study. Fertil Steril 116(6): 1568-1577.
- Taniguchi F, Ota I, Iba Y, Toda T, Tagashira Y, et al., (2019) The efficacy and safety of dydrogesterone for treatment of dysmenorrhea: An open-label multicenter clinical study. J Obstet Gynaecol Res 45(1): 168-175.
- Griffiths MJ, Horne AW, Gibson DA, Roberts N, Saunders PTK, et al., (2024) Endometriosis: recent advances that could accelerate diagnosis and improve care. Trends Mol Med 30(9): 875-889.
- Li M, H Chang (2025) Research Progress on the Correlation Between Microecological Flora and Endometriosis. Medical Journal of Peking Union Medical College Hospital 16(1): 184-191.